Summary
Clinical characteristics.
Fructose-1,6-bisphosphatase (FBP1) deficiency is characterized by episodic acute crises of lactic acidosis and ketotic hypoglycemia, manifesting as hyperventilation, apneic spells, seizures, and/or coma. Acute crises are most common in early childhood; nearly half of affected children have hypoglycemia in the neonatal period (especially the first 4 days) resulting from deficient glycogen stores. Factors known to trigger episodes include fever, fasting, decreased oral intake, vomiting, infections, and ingestion of large amounts of fructose.
In untreated individuals, symptoms worsen progressively as continued catabolism leads to multiorgan failure (especially liver, brain, and later heart). Morbidity and mortality are high. Sepsis, blindness, and Reye syndrome-like presentation have been reported.
In between acute episodes, children are asymptomatic. While the majority of affected children have normal growth and psychomotor development, a few have intellectual disability, presumably due to early and prolonged hypoglycemia.
Diagnosis/testing.
The diagnosis of FBP1 deficiency is established in a proband with suggestive clinical and metabolic findings by identification of EITHER biallelic FBP1 pathogenic variants on molecular genetic testing OR deficient fructose-1,6-bisphosphatase 1 (FBP1) activity in liver or mononuclear white blood cells. Molecular genetic testing is generally preferred because of its widespread availability and accuracy.
Management.
Treatment of manifestations: Intervention (oral or IV glucose) should take place early in an acute crisis while the blood glucose is normal due to the possibility of delayed hypoglycemia, which only occurs relatively late in the course of acute metabolic decompensation.
The mainstay of routine daily management is prevention of hypoglycemia by avoiding fasting (including use of uncooked cornstarch overnight), consuming frequent meals, and appropriate management of acute intercurrent illnesses.
Prevention of primary manifestations: Routine daily management to prevent hypoglycemia, attention to agents/circumstances to avoid, and routine immunizations, including annual influenza vaccine to reduce the risk of infection, which can precipitate hypoglycemia.
Surveillance: Long-term monitoring of developmental milestones in affected children and quality of life issues for affected individuals and their parents/caregivers; monitoring for excessive weight gain at each visit.
Agents/circumstances to avoid: Food items or medicines that contain fructose, sucrose, glycerol, and/or sorbitol, especially during acute crisis in infancy or early childhood. Although small amounts of fructose (≤2 g/kg/day) are generally well tolerated, single ingestion of high dose of fructose (>1g/kg) is harmful, especially in younger children. Fructose tolerance testing ("fructose challenge") to diagnose FBP1 deficiency can be hazardous and should not be performed.
Evaluation of relatives at risk: When the familial FBP1 variants are known, it is appropriate to clarify the genetic status of apparently asymptomatic older and younger at-risk sibs of an affected individual in order to identify as early as possible those who would benefit from prompt initiation of preventive measures.
Pregnancy management: For a pregnant woman with FBP1 deficiency, consider referral to a high-risk obstetric center and consultation with a metabolic physician. Home glucose monitoring and consumption of uncooked cornstarch at night as needed as carbohydrate requirements increase during pregnancy. During labor, continuous glucose infusion is recommended to maintain euglycemia.
Genetic counseling.
FBP1 deficiency is inherited in an autosomal recessive manner. When both parents are known to be heterozygous for an FBP1 pathogenic variant, each sib of an affected individual has a 25% chance of being affected, a 50% chance of being an asymptomatic carrier, and a 25% chance of being unaffected and not a carrier. Once the FBP1 pathogenic variants have been identified in an affected family member, carrier testing for at-risk relatives, prenatal testing for a pregnancy at increased risk, and preimplantation genetic testing are possible.
Diagnosis
Formal diagnostic criteria for fructose-1,6-bisphosphatase (FBP1) deficiency have not been established.
Suggestive Findings
FBP1 deficiency should be suspected in individuals with the following characteristic clinical features and metabolic findings.
Clinical features
- Episodes of acute crisis may manifest as hyperventilation, apneic spells, seizures, and/or coma, most commonly in neonates and infants. The course of illness is precipitous and may be lethal, especially in neonates and infants.
- Other features can include episodic irritability, tachycardia, hypotonia, and hyperhidrosis.
- Hepatomegaly is common.
- Inter-crisis periods are uneventful with normal development and growth.
Metabolic findings. A strong clinical suspicion should arise in individuals with recurrent episodes of ketotic hypoglycemia with lactic acidosis and urinary organic acid analysis showing a glycerol and glycerol-3-phosphate peak. Although this combination is fairly specific for FBP1 deficiency, establishing the diagnosis requires enzyme assay and/or molecular genetic testing (see Establishing the Diagnosis).
- Hypoglycemia (plasma glucose <40mg/dL in neonates; <60 mg/dL in older infants, children, and adults; reference range 70-120 mg/dL)
- High anion-gap metabolic acidosis
- Lactic acidemia (plasma lactate >2.5 mmol/L; reference range: 0.5-2.2 mmol/L) with possible elevated lactate:pyruvate ratio and hyperalaninemia.
- Ketosis (individuals with low ketones have been reported)
- Elevated glycerol 3-phosphate is an important biomarker for this disorder on urine organic acid analysis. Glycerol 3-phosphate is more specific as glycerol is also elevated in glycerol kinase deficiency (see NROB1-Related Adrenal Hypoplasia Congenita) [Nakai et al 1993]. It is important to collect the urine sample in the period of crisis, as the glycerol 3-phosphate level returns to normal once the individual is in the well state [Moey et al 2018].
- Pseudo-hypertriglyceridemia. Although plasma triglycerides are commonly increased, it is not the triglycerides but the glycerol levels that are high (often referred to as "pseudo-hypertriglyceridemia"). The commonly available biochemical assays for plasma triglycerides cannot differentiate glycerol from triglycerides, and hence overestimate the triglyceride levels due to the high concentration of glycerol in plasma [Afroze et al 2013]. Glycerol blanking methods can be used to measure the true levels of triglycerides [Cole 1990].
- Hyperuricemia (plasma uric acid >5.0 mg/dL; reference range 2.0-5.0 mmol/L
- Increased free fatty acids (in some cases)
Establishing the Diagnosis
The diagnosis of fructose-1,6-bisphosphatase 1 deficiency is established in a proband with suggestive clinical and metabolic findings by identification of EITHER biallelic FBP1 pathogenic (or likely pathogenic) variants on molecular genetic testing (Table 1) OR deficient fructose-1,6-bisphosphatase 1 (FBP1) activity in liver or mononuclear white blood cells. Molecular genetic testing is generally preferred because of its widespread availability and accuracy.
Note: (1) Per ACMG/AMP variant interpretation guidelines, the terms "pathogenic variants" and "likely pathogenic variants" are synonymous in a clinical setting, meaning that both are considered diagnostic and both can be used for clinical decision making [Richards et al 2015]. Reference to "pathogenic variants" in this section is understood to include any likely pathogenic variants. (2) Identification of biallelic FBP1 variants of uncertain significance (or of one known FBP1 pathogenic variant and one FBP1 variant of uncertain significance) does not establish or rule out the diagnosis.
Molecular Genetic Testing
When the clinical and metabolic findings suggest the diagnosis of fructose-1,6-bisphosphatase deficiency, molecular genetic testing approaches can include single-gene testing or use of a multigene panel:
- Single-gene testing. Sequence analysis of FBP1 detects small intragenic deletions/insertions and missense, nonsense, and splice site variants; typically, exon or whole-gene deletions/duplications are not detected. Perform sequence analysis first. If only one or no pathogenic variant is found, perform gene-targeted deletion/duplication analysis to detect intragenic deletions or duplications.Note: Pathogenic variants reported frequently in individuals of certain ancestries are noted in Table 7. Given the rarity of fructose-1-6-bisphosphatase deficiency, the efficacy of targeted testing for specific pathogenic variants in individuals of these ancestries is not known.
- A multigene panel (such as one for hypoglycemia or lactic acidosis) that includes FBP1 and other genes of interest (see Differential Diagnosis) is most likely to identify the genetic cause of the condition while limiting identification of variants of uncertain significance and pathogenic variants in genes that do not explain the underlying phenotype. Note: (1) The genes included in the panel and the diagnostic sensitivity of the testing used for each gene vary by laboratory and are likely to change over time. (2) Some multigene panels may include genes not associated with the condition discussed in this GeneReview. Of note, given the rarity of fructose-1,6-bisphosphatase deficiency, some panels may not include FBP1. (3) In some laboratories, panel options may include a custom laboratory-designed panel and/or custom phenotype-focused exome analysis that includes genes specified by the clinician. (4) Methods used in a panel may include sequence analysis, deletion/duplication analysis, and/or other non-sequencing-based tests. For this disorder a multigene panel that also includes deletion/duplication analysis is recommended (see Table 1).
Table 1.
Molecular Genetic Testing Used in Fructose-1,6-Bisphosphatase Deficiency
Analysis of Fructose-1,6-Bisphosphatase (FBP1 or FBPase) Activity
While enzymatic activity in leukocytes and liver is very specific, testing is not widely available [Besley et al 1994, Lebigot et al 2015]: spectrophotometric analysis to estimate enzyme activity in leukocytes is performed in a few laboratories worldwide. The usual assay tests FBP1 activity by measuring the amount of NADPH formed – a nonspecific assay as NADPH may be formed from other reactions within the cells. Specificity can be achieved by testing for FBP1 activity in the presence and absence of AMP, a specific inhibitor for the enzyme [Lebigot et al 2015]. FBP1 stability in untreated blood samples at +4 °C lasts 24 hours, and therefore samples can be transported long distances to a reference laboratory.
Clinical Characteristics
Clinical Description
The manifestations of fructose-1,6-bisphosphatase (FBP1) deficiency are generally episodic due to lactic acidosis and ketotic hypoglycemia, which are often triggered by fasting or febrile infections. The episodes of acute crisis are most frequent in early life – neonatal period, infancy, and early childhood – and subsequently decrease in frequency. In FBP1 deficiency, several metabolic derangements can occur with or without hypoglycemia.
Classically, FBP1 deficiency manifests in the first year of life. Nearly half of affected infants present within the first four days of life with an acute crisis. Neonatal presentation results from hypoglycemia due to deficient glycogen stores [Steinmann & Santer 2016].
Acute crises are characterized by episodes of hyperventilation, apneic spells, seizures, and/or coma, often with hepatomegaly. Muscular hypotonia may also be present. This may be associated with transient liver dysfunction (transaminitis), which does not require specific treatment [Bhai et al 2018]. With age, the frequency of attacks decreases and episodes are characterized by irritability, somnolence, hypotonia, and dyspnea.
Factors known to trigger episodes include fever, fasting, decreased oral intake, vomiting, infections, and ingestion of large amounts of fructose. Episodes tend to be recurrent. Often four to five episodes occur before the correct diagnosis is established [Lebigot et al 2015].
In between crises, children are asymptomatic and the majority experience normal growth and psychomotor development [Steinmann & Santer 2016]. A few children with brain injury and/or intellectual disability have been reported, probably related to early and prolonged hypoglycemia [Li et al 2017, Moey et al 2018].
In untreated individuals, symptoms worsen progressively as continued catabolism leads to multiorgan failure (especially liver, brain, and later heart). Morbidity and mortality are high. Sepsis, blindness, and Reye syndrome-like presentation have been reported [Lebigot et al 2015, Bhai et al 2018].
Genotype-Phenotype Correlations
No genotype-phenotype correlations have been identified.
Nomenclature
Baker & Winegrad [1970] first described deficiency of hepatic fructose-1,6-bisphosphatase 1 (hence the eponymous Baker-Winegrad disease) in a child with hypoglycemia and metabolic acidosis on fasting.
Prevalence
Fructose-1,6-bisphosphatase deficiency is rare. Approximately 150 affected individuals have been reported to date.
An estimated prevalence of fructose-1,6-bisphosphatase deficiency is 1:350,000 in the Dutch population [Visser et al 2004] and <1:900,000 in the French population [Lebigot et al 2015]. The disorder may be more frequent in populations with higher rates of consanguinity [Santer et al 2016].
Genetically Related (Allelic) Disorders
No phenotypes other than those discussed in this GeneReview are known to be associated with germline pathogenic variants in FBP1.
Differential Diagnosis
Table 2a provides a comparative analysis of disorders with clinical similarities to fructose-1,6-bisphosphatase (FBP1) deficiency. Table 2b compares the biochemical parameters of these disorders.
Information on mitochondrial respiratory chain and Krebs cycle disorders and fatty acid oxidation defects follows Table 2b.
Table 2a.
Disorders (and Associated Genes) of Interest in the Differential Diagnosis of Fructose-1,6-Bisphosphatase Deficiency
Table 2b.
Biochemical Parameters of Disorders in the Differential Diagnosis of Fructose-1,6-Bisphosphatase Deficiency
Fatty acid oxidation defects (FAODs) are mitochondrial disorders caused by defective beta-oxidation of fatty acids. FAODs can present in neonates with hypoglycemia, hyperammonemia, and reduced/absent ketones. As ketones could be normal in FBP1 deficiency, diagnosis of FAOD is based on elevated acylcarnitine profiles on MS/MS.
Mitochondrial respiratory chain and Krebs cycle disorders. Mitochondrial respiratory chain disorders are a heterogeneous group of disorders that share involvement of the cellular bioenergetic machinery due to molecular defects affecting the mitochondrial oxidative phosphorylation system. The variable presentation can include neonatal metabolic acidosis with increased lactate. The usual disease course is a gradually progressive loss of developmental milestones; however, more rapid decline occurs with episodic crises. Urine organic acid profile may reveal distinctive elevation of fumaric acid or other Krebs cycle intermediates, reflecting the site of the enzyme deficiency. See Mitochondrial Disorders Overview.
Management
Evaluations Following Initial Diagnosis
To establish the extent of disease and needs in a child diagnosed with fructose-1,6-bisphosphatase (FBP1) deficiency who is not in acute crisis, the evaluations summarized in Table 3 (if not performed as part of the evaluation that led to the diagnosis) are recommended [Wang et al 2017, Bhai et al 2018].
Note that the management of possible multisystem complications resulting from early and prolonged hypoglycemia are not discussed in this GeneReview.
Table 3.
Recommended Evaluations Following Initial Diagnosis of Fructose-1,6-Bisphosphatase Deficiency for a Child Not in Acute Crisis
Treatment of Manifestations
An international survey of 126 patients from 36 centers revealed widely varying practices of fructose/sucrose restriction; the authors concluded that internationally accepted guidelines for management and surveillance were needed [Pinto et al 2018].
Management of acute crises. Acute crises are more frequent in early life – neonatal period, infancy, and early childhood – with gradual decrease afterwards.
Intervention (oral glucose or IV dextrose) should take place early in an acute crisis while the blood glucose is normal due to the possibility of delayed hypoglycemia, which only occurs relatively late in the course of acute metabolic decompensation.
The family must be given the contact information for an expert metabolic center and a clear emergency outpatient treatment plan during illness (Table 4) and emergency letters/cards with information on principles of acute in-patient treatment (Table 5).
Emergency outpatient treatment. The threshold for emergency outpatient evaluation and treatment should be low. Acute management guidelines are available from the British Inherited Metabolic Disease Group. See protocol (pdf).
Laboratory evaluations during acute illnesses should include pH and blood gases, glucose (laboratory and bedside strip test), urea and electrolytes, complete blood count, lactate, blood culture, and urine ketones.
The child should receive emergency treatment if the child is vomiting, complaining of stomach pain, or not wanting to eat or drink, irrespective of associated symptoms (e.g., fever, cough, cold).
Table 4.
Emergency Outpatient Treatment in Individuals with Fructose-1,6-Bisphosphatase Deficiency
Acute in-patient treatment. Acute manifestations of hypoglycemia (lethargy, encephalopathy, seizures, hyperhidrosis), often occurring in the setting of intercurrent illness and/or inadequate caloric intake, should be managed symptomatically and with generous caloric support in a hospital setting, with aggressive treatment and supportive care of any identified or clinically suspected acute conditions.
Table 5.
Acute In-Patient Treatment in Individuals with Fructose-1,6-Bisphosphatase Deficiency
Prevention of Primary Manifestations
The main principles of routine daily management are the following:
- Prevention of hypoglycemia (by avoiding fasting and consuming frequent meals). Slowly absorbed carbohydrates such as uncooked cornstarch (1-2 g/kg) mixed in milk or water at bedtime may be introduced to children after age six to 12 months who have nocturnal hypoglycemia [Pinto et al 2018].
- Appropriate management of acute intercurrent illnesses, which can exacerbate the need for glucose
- Restriction of food items or medicines that contain fructose, sucrose, glycerol, and/or sorbitol
Also advised are routine immunizations, including annual influenza vaccine, to reduce the risk of infections that can precipitate hypoglycemia.
Surveillance
No formal guidelines for long-term surveillance for individuals with FBP1 deficiency exist. Table 6 summarizes long-term surveillance based on the natural history of the disorder.
Table 6.
Recommended Long-Term Surveillance for Individuals with Fructose-1,6-Bisphosphatase Deficiency
Agents/Circumstances to Avoid
Avoid ingestion of food items or medicines that contain fructose, sucrose, glycerol, and/or sorbitol, especially during acute crises in infancy or early childhood.
Although small amounts of fructose (≤2 g/kg/day) are well tolerated by individuals with FBP1 deficiency, single ingestion of high doses of fructose (>1g/kg) is harmful, especially in younger children.
The fructose tolerance testing ("fructose challenge") to diagnose FBP1 deficiency can be hazardous and should not be performed.
Evaluation of Relatives at Risk
It is appropriate to clarify the status of apparently asymptomatic older and younger at-risk sibs of an affected individual in order to identify as early as possible those who would benefit from prompt initiation of measures to prevent acute crises (frequent feedings and avoidance of fasting) and prompt treatment of infections / febrile illnesses. Evaluations should include targeted molecular genetic testing if the FBP1 pathogenic variants in the family are known.
For newborns who may be at risk for FBP1 deficiency, it is important to avoid fasting, to feed regularly, and to monitor for hypoglycemia and acidosis while awaiting results of biochemical and/or molecular genetic testing. Monitoring for hypoglycemia can include use of bedside glucometers and clinical monitoring for lethargy, poor intake, vomiting, hypothermia, and/or tachypnea. A low threshold for starting an intravenous infusion of 5%-10% dextrose may help prevent triggering of an acute crisis, especially in newborns who have low birth weight, are preterm, or have a diabetic mother.
See Genetic Counseling for issues related to testing of at-risk relatives for genetic counseling purposes.
Pregnancy Management
Although successful pregnancies have been reported in women with FBP1 deficiency [Åsberg et al 2010], certain precautions should be taken:
- Pre-pregnancy
- Counseling that emphasizes the importance of frequent eating in preventing hypoglycemia and the use of blood glucose monitoring for early detection of hypoglycemia
- Consider referral to a high-risk obstetric center and consultation with a metabolic physician.
- During pregnancy
- Close home glucose monitoring for early detection of hypoglycemia
- Maintenance of glycemic control by taking uncooked cornstarch at night as needed [Krishnamurthy et al 2007, Sugita et al 2014]
- In the third trimester when the physiologic requirements of energy are highest, increase in dietary intake and glucose monitoring as needed
- During labor. Continuous glucose infusion to maintain euglycemia
Therapies Under Investigation
Search ClinicalTrials.gov in the US and EU Clinical Trials Register in Europe for access to information on clinical studies for a wide range of diseases and conditions. Note: There may not be clinical trials for this disorder.
Genetic Counseling
Genetic counseling is the process of providing individuals and families with information on the nature, mode(s) of inheritance, and implications of genetic disorders to help them make informed medical and personal decisions. The following section deals with genetic risk assessment and the use of family history and genetic testing to clarify genetic status for family members; it is not meant to address all personal, cultural, or ethical issues that may arise or to substitute for consultation with a genetics professional. —ED.
Mode of Inheritance
Fructose-1,6-bisphosphatase (FBP1) deficiency is inherited in an autosomal recessive manner.
Parents of a proband
- The parents of an affected child are obligate heterozygotes (i.e., presumed to be carriers of one FBP1 pathogenic variant based on family history).
- Molecular genetic testing is recommended for the parents of a proband to confirm that both parents are heterozygous for an FBP1 pathogenic variant and to allow reliable recurrence risk assessment. (Although a de novo pathogenic variant has not been reported in FBP1 deficiency to date, de novo variants are known to occur at a low but appreciable rate in autosomal recessive disorders [Jónsson et al 2017].)
- Heterozygotes (carriers) are asymptomatic and are not at risk of developing the disorder.
Sibs of a proband
- If both parents are known to be heterozygous for an FBP1 pathogenic variant, each sib at conception has a 25% chance of being affected, a 50% chance of being an asymptomatic carrier, and a 25% chance of being unaffected and not a carrier.
- Heterozygotes (carriers) are asymptomatic and are not at risk of developing the disorder.
Offspring of a proband. Unless an affected individual's reproductive partner also has FBP1 deficiency or is a carrier, offspring will be obligate heterozygotes (carriers) for one of the FBP1 pathogenic variants identified in the proband.
Other family members. Each sib of the proband's parents is at a 50% risk of being a carrier of an FBP1 pathogenic variant.
Carrier Detection
Molecular genetic testing. Carrier testing for at-risk relatives requires prior identification of the FBP1 pathogenic variants in the family.
Note: Biochemical enzyme-based testing is not reliable for carrier detection.
Related Genetic Counseling Issues
See Management, Evaluation of Relatives at Risk for information on evaluating at-risk relatives for the purpose of early diagnosis and treatment.
Family planning
- The optimal time for determination of genetic risk, clarification of carrier status, and discussion of the availability of prenatal/preimplantation genetic testing is before pregnancy.
- It is appropriate to offer genetic counseling (including discussion of potential risks to offspring and reproductive options) to young adults who are affected, are carriers, or are at risk of being carriers.
DNA banking. Because it is likely that testing methodology and our understanding of genes, pathogenic mechanisms, and diseases will improve in the future, consideration should be given to banking DNA from probands in whom a molecular diagnosis has not been confirmed (i.e., the causative pathogenic mechanism is unknown). For more information, see Huang et al [2022].
Prenatal Testing and Preimplantation Genetic Testing
Molecular genetic testing. Once the FBP1 pathogenic variants have been identified in an affected family member, prenatal and preimplantation genetic testing are possible.
Note: Biochemical testing is not a reliable method for prenatal diagnosis as FBP1 enzyme activity has been reported to be low in human placenta [Papamarcaki & Tsolas 1991].
Differences in perspective may exist among medical professionals and within families regarding the use of prenatal testing. While most centers would consider use of prenatal testing to be a personal decision, discussion of these issues may be helpful.
Resources
GeneReviews staff has selected the following disease-specific and/or umbrella support organizations and/or registries for the benefit of individuals with this disorder and their families. GeneReviews is not responsible for the information provided by other organizations. For information on selection criteria, click here.
- Metabolic Support UKUnited KingdomPhone: 0845 241 2173
Molecular Genetics
Information in the Molecular Genetics and OMIM tables may differ from that elsewhere in the GeneReview: tables may contain more recent information. —ED.
Table A.
Fructose-1,6-Bisphosphatase Deficiency: Genes and Databases
Table B.
OMIM Entries for Fructose-1,6-Bisphosphatase Deficiency (View All in OMIM)
Molecular Pathogenesis
Fructose-1,6-bisphosphatase 1 (FBPase, or FBP1) is a key enzyme of the gluconeogenic pathway. Its deficiency impairs glucose production from all gluconeogenic precursors, including dietary fructose. As a result, euglycemia in persons with FBP1 deficiency depends on glycogen stores in the liver. During glycemic stress (including catabolic states such as infections and fasting), glycogen stores are depleted and the gluconeogenic substrates pyruvate, alanine, and glycerol accumulate (Figure 1). The subsequent conversion of pyruvate into lactate and acetyl CoA results in lactic acidemia and ketosis.
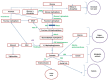
Figure 1:
Pathophysiology of FBP1 deficiency
Deficient conversion of glucose-1-phosphate to glucose also activates the pentose phosphate pathway, causing both the production of ribose-5-phosphate and the synthesis of purines and pyrimidines. The subsequent breakdown of purines and pyrimidines results in hyperuricemia.
Mechanism of disease causation. Loss of fructose-1,6-bisphosphatase 1 function
FBP1-specific laboratory technical considerations. FBP1 comprises one noncoding exon (exon 1) and seven coding exons; it spans more than 31 kb [el-Maghrabi et al 1995]. Deletions of exons 2, 3, 3-7, and 8 as well as whole-gene deletions have been reported.
(The six FBP1 introns are in the same positions in the rat gene.)
Table 7.
Notable FBP1 Pathogenic Variants
Chapter Notes
Acknowledgments
The authors would like to acknowledge our senior colleagues, Dr Ishwar Chander Verma, Dr Ratna D Puri, and Dr Renu Saxena, for rendering support in writing this article. Significant contributions from other colleagues, Dr Pratibha Bhai, Dr Deepti Gupta, Dr Ranjana Mishra, Dr Vibha Jain, and Dr Jyotsna Verma, are also gratefully acknowledged.
Revision History
- 5 December 2019 (bp) Review posted live
- 20 May 2019 (sbm) Original submission
References
Literature Cited
- Afroze B, Yunus Z, Steinmann B, Santer R. Transient pseudo-hypertriglyceridemia: a useful biochemical marker of fructose-1,6-bisphosphatase deficiency. Eur J Pediatr. 2013;172:1249–53. [PubMed: 23881342]
- Åsberg C, Hjalmarson O, Alm J, Martinsson T, Waldenström J, Hellerud C. Fructose 1,6-bisphosphatase deficiency: enzyme and mutation analysis performed on calcitriol-stimulated monocytes with a note on long-term prognosis. J Inherit Metab Dis. 2010;33:S113–21. [PubMed: 20151204]
- Baker L, Winegrad AI. Fasting hypoglycaemia and metabolic acidosis associated with deficiency of hepatic fructose-1,6-diphosphatase activity. Lancet. 1970;2:13–6. [PubMed: 4193749]
- Besley GT, Walter JH, Lewis MA, Chard CR, Addison GM. Fructose-1,6-bisphosphatase deficiency: severe phenotype with normal leukocyte enzyme activity. J Inherit Metab Dis. 1994;17:333–5. [PubMed: 7807945]
- Bhai P, Bijarnia-Mahay S, Puri RD, Saxena R, Gupta D, Kotecha U, Sachdev A, Gupta D, Vyas V, Agarwal D, Jain V, Bansal RK, Kumar TG, Verma IC. Clinical and molecular characterization of Indian patients with fructose-1, 6-bisphosphatase deficiency: identification of a frequent variant (E281K). Ann Hum Genet. 2018;82:309–17. [PubMed: 29774539]
- Cole TG. Glycerol blanking in triglyceride assays: is it necessary? Clin Chem. 1990;36:1267–8. [PubMed: 2136327]
- el-Maghrabi MR, Lange AJ, Jiang W, Yamagata K, Stoffel M, Takeda J, Fernald AA, Le Beau MM, Bell GI, Baker L, et al. Human fructose-1,6-bisphosphatase gene (FBP1): exon-intron organization, localization to chromosome bands 9q22.2-q22.3, and mutation screening in subjects with fructose-1,6-bisphosphatase deficiency. Genomics. 1995;27:520–5. [PubMed: 7558035]
- Herzog B, Morris AA, Saunders C, Eschrich K. Mutation spectrum in patients with fructose-1,6-bisphosphatase deficiency. J Inherit Metab Dis. 2001;24:87–8. [PubMed: 11286391]
- Huang SJ, Amendola LM, Sternen DL. Variation among DNA banking consent forms: points for clinicians to bank on. J Community Genet. 2022;13:389–97. [PMC free article: PMC9314484] [PubMed: 35834113]
- Ijaz S, Zahoor MY, Imran M, Ramzan K, Bhinder MA, Shakeel H, Iqbal M, Aslam A, Shehzad W, Cheema HA, Rehman H. Genetic analysis of fructose-1,6-bisphosphatase (FBPase) deficiency in nine consanguineous Pakistani families. J Pediatr Endocrinol Metab. 2017;30:1203–10. [PubMed: 29016355]
- Jónsson H, Sulem P, Kehr B, Kristmundsdottir S, Zink F, Hjartarson E, Hardarson MT, Hjorleifsson KE, Eggertsson HP, Gudjonsson SA, Ward LD, Arnadottir GA, Helgason EA, Helgason H, Gylfason A, Jonasdottir A, Jonasdottir A, Rafnar T, Frigge M, Stacey SN, Th Magnusson O, Thorsteinsdottir U, Masson G, Kong A, Halldorsson BV, Helgason A, Gudbjartsson DF, Stefansson K. Parental influence on human germline de novo mutations in 1,548 trios from Iceland. Nature. 2017;549:519–22. [PubMed: 28959963]
- Kikawa Y, Inuzuka M, Jin BY, Kaji S, Koga J, Yamamoto Y, Fujisawa K, Hata I, Nakai A, Shigematsu Y, Mizunuma H, Taketo A, Mayumi M, Sudo M. Identification of genetic mutations in Japanese patients with fructose-1,6-bisphosphatase deficiency. Am J Hum Genet. 1997;61:852–61. [PMC free article: PMC1715983] [PubMed: 9382095]
- Krishnamurthy V, Eschrich K, Boney A, Sullivan J, McDonald M, Kishnani PS, Koeberl DD. Three successful pregnancies through dietary management of fructose-1,6-bisphosphatase deficiency. J Inherit Metab Dis. 2007;30:819. [PubMed: 17705024]
- Lebigot E, Brassier A, Zater M, Imanci D, Feillet F, Thérond P, de Lonlay P, Boutron A. Fructose 1,6-bisphosphatase deficiency: clinical, biochemical and genetic features in French patients. J Inherit Metab Dis. 2015;38:881–7. [PubMed: 25601412]
- Lee H, Kwon A, Kim HS, Lee JS. Fructose-1,6-bisphosphatase deficiency presented with complex febrile convulsion. Neuro Endocrinol Lett. 2019;39:533–6. [PubMed: 30927757]
- Li N, Chang G, Xu Y, Ding Y, Li G, Yu T, Qing Y, Li J, Shen Y, Wang J, Wang X. Clinical and molecular characterization of patients with fructose 1,6-bisphosphatase deficiency. Int J Mol Sci. 2017;18:E857. [PMC free article: PMC5412439] [PubMed: 28420223]
- Moey LH, Abdul Azize NA, Yakob Y, Leong HY, Keng WT, Chen BC, Ngu LH. Fructose-1,6-bisphosphatase deficiency as a cause of recurrent hypoglycemia and metabolic acidosis: clinical and molecular findings in Malaysian patients. Pediatr Neonatol. 2018;59:397–403. [PubMed: 29203193]
- Nakai A, Shigematsu Y, Liu YY, Kikawa Y, Sudo M. Urinary sugar phosphates and related organic acids in fructose-1,6-diphosphatase deficiency. J Inherit Metab Dis. 1993;16:408–14. [PubMed: 8412001]
- Papamarcaki T, Tsolas O. Demonstration of fructose 1,6-bisphosphatase in human term placenta. Experientia. 1991;47:67–8. [PubMed: 1847882]
- Pinheiro FC, Sperb-Ludwig F, Ligabue-Braun R, Schüler-Faccini L, de Souza CFM, Vairo F, Schwartz IVD. Genetic analysis of patients with fructose-1,6-bisphosphatase deficiency. Gene. 2019;699:102–9. [PubMed: 30858132]
- Pinto A, Alfadhel M, Akroyd R, Atik Altınok Y, Bernabei SM, Bernstein L, Bruni G, Caine G, Cameron E, Carruthers R, Cochrane B, Daly A, de Boer F, Delaunay S, Dianin A, Dixon M, Drogari E, Dubois S, Evans S, Gribben J, Gugelmo G, Heidenborg C, Hunjan I, Kok IL, Kumru B, Liguori A, Mayr D, Megdad E, Meyer U, Oliveira RB, Pal A, Pozzoli A, Pretese R, Rocha JC, Rosenbaum-Fabian S, Serrano-Nieto J, Sjoqvist E, Timmer C, White L, van den Hurk T, van Rijn M, Zweers H, Ziadlou M, MacDonald A. International practices in the dietary management of fructose 1-6 biphosphatase deficiency. Orphanet J Rare Dis. 2018;13:21. [PMC free article: PMC5785792] [PubMed: 29370874]
- Prahl P, Christensen E, Hansen L, Mortensen HB. Fructose 1,6-bisphosphatase deficiency as a cause of recessive serious hypoglycaemia. Ugeskr Laeger. 2006;168:4014–5. [PubMed: 17125659]
- Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, Grody WW, Hegde M, Lyon E, Spector E, Voelkerding K, Rehm HL, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405–24. [PMC free article: PMC4544753] [PubMed: 25741868]
- Santer R, du Moulin M, Shahinyan T, Vater I, Maier E, Muntau AC, Steinmann B. A summary of molecular genetic findings in fructose-1,6-bisphosphatase deficiency with a focus on a common long-range deletion and the role of MLPA analysis. Orphanet J Rare Dis. 2016;11:44. [PMC free article: PMC4839065] [PubMed: 27101822]
- Steinmann B, Santer R. Disorders of fructose metabolism. In: Saudubray JM, ed. Inborn Metabolic Diseases. 6 ed. Springer-Verlag; 2016:165–7.
- Sugita G, Tsuyoshi H, Nishijima K, Yoshida Y. Fructose-1,6-bisphosphatase deficiency: a case of a successful pregnancy by closely monitoring metabolic control. JIMD Rep. 2014;14:115–8. [PMC free article: PMC4213334] [PubMed: 24470127]
- Takagi D, Ben-Ari J, Nemet D, Zeharia A, Eliakim A. Recurrent infantile hypoglycemia due to combined fructose-1,6-diphosphatase deficiency and growth hormone deficiency. J Pediatr Endocrinol Metab. 2013;26:761–3. [PubMed: 23585210]
- Visser G, Bakker HD, de Klerk JBC, Smeitink JAM, Smit GPA, Wijburg FA. Natural history and treatment of fructose-1,6,-bisphosphatase deficiency in the Netherlands. J Inherit Metab Dis. 2004;27:207. (abstract)
- Wang S, Sun J, Kong X, Yang C, Feng Z. Fructose 1,6-bisphosphatase deficiency: clinical manifestations and genetic features in two Chinese patients. Int J Clin Exp Med. 2017;10:7273–8.
Publication Details
Author Information and Affiliations
New Delhi, India
New Delhi, India
New Delhi, India
Publication History
Initial Posting: December 5, 2019.
Copyright
GeneReviews® chapters are owned by the University of Washington. Permission is hereby granted to reproduce, distribute, and translate copies of content materials for noncommercial research purposes only, provided that (i) credit for source (http://www.genereviews.org/) and copyright (© 1993-2024 University of Washington) are included with each copy; (ii) a link to the original material is provided whenever the material is published elsewhere on the Web; and (iii) reproducers, distributors, and/or translators comply with the GeneReviews® Copyright Notice and Usage Disclaimer. No further modifications are allowed. For clarity, excerpts of GeneReviews chapters for use in lab reports and clinic notes are a permitted use.
For more information, see the GeneReviews® Copyright Notice and Usage Disclaimer.
For questions regarding permissions or whether a specified use is allowed, contact: ude.wu@tssamda.
Publisher
University of Washington, Seattle, Seattle (WA)
NLM Citation
Bijarnia-Mahay S, Bhatia S, Arora V. Fructose-1,6-Bisphosphatase Deficiency. 2019 Dec 5. In: Adam MP, Feldman J, Mirzaa GM, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2024.
