Abstract
We show that the P2Y6 receptor, a G-protein-coupled receptor with high affinity for the nucleotide uridine diphosphate, is an important endogenous inhibitor of T cell function in allergic pulmonary inflammation. Mice conditionally deficient in P2Y6 receptors [p2ry6 (flox/flox);cre/+ mice] exhibited severe airway and tissue pathology relative to P2Y6-sufficient [p2ry6 (flox/flox)] littermates (+/+ mice) when treated intranasally with an extract (Df) of the dust mite Dermatophagoides farinae. P2Y6 receptors were inducibly expressed by lung, lymph node and splenic CD4+ and CD8+ T cells of Df-treated +/+ mice. Df-restimulated P2Y6-deficient lymph node cells produced higher levels of Th1 and Th2 cytokines, and polyclonally-stimulated P2Y6-deficient CD4+ T cells proliferated faster than comparably stimulated P2Y6-sufficient cells. The absence of P2Y6 receptors on CD4+ cells, but not antigen presenting cells, was sufficient to amplify cytokine generation. Thus, P2Y6 receptors protect the lung against exuberant allergen-induced pulmonary inflammation by inhibiting the activation of effector T cells.
Introduction
Nucleotides, the structural subunits of the nucleic acids, are also important extracellular signaling molecules (1,2). They are stored in cytosolic and secretory compartments of cells, and are released in response to cell injury, hypoxia, shear stress, and tissue injury (3–5). Nucleotides are also released by platelets, endothelial cells, mast cells (MCs), macrophages, and T cells in response to physiologic activation (6–9). Consequently, extracellular nucleotides accumulate at sites of vascular injury, hypoxia, thrombosis, inflammation, and immune cell activation. Both adenine-containing (ATP, ADP) and uracil-containing UTP, UDP) nucleotides have extracellular functions mediated by cognate cell surface receptors. These receptors fall into two classes; P2X receptors, which are ligand-gated ion channels that mediate calcium and potassium fluxes in response to ATP, and P2Y receptors, which are G protein-coupled receptors (GPCRs2) (10–12). Although adenine nucleotides and their receptors play established roles in platelet aggregation (13), pain perception (14), and cellular responses to hypoxic injury (15), the functions of many P2Y receptors in vivo remain unknown.
P2Y6 receptors are the only known high affinity receptors selective for UDP. They are expressed on both hematopoietic (macrophages, dendritic cells, MCs, T cells), and non-hematopoietic (vascular smooth muscle, epithelium, endothelium) cell types (16–25). Most functions attributed to P2Y6 receptors in vitro suggest a role in modulating cellular responses to inflammation through autocrine or paracrine actions of uracil nucleotides. Endogenous uracil nucleotides amplify airway epithelial chemokine production in response to neutrophil-derived peptides through P2Y6 receptors in vitro (26). P2Y6 receptors are strongly expressed by colonic epithelial cells in biopsies from patients with inflammatory bowel disease, and mediate production of interleukin (IL)-8 by colonic epithelial cell lines in vitro (19). P2Y6 receptors amplify chemokine generation by a lipopolysaccharide (LPS)-stimulated monocytic cell line (27), amplify endothelial cell activation in response to LPS (28), and enhance the release of macrophage inflammatory protein-1β by human MCs stimulated with leukotriene D4 (20). Human T cells express P2Y6 receptors after activation in vitro and at sites of inflammation in vivo (23), and pharmacologic blockade of P2Y6 receptors inhibits proliferation, CD25 expression, and generation of cytokines by mouse T cells in response to polyclonal and antigen-specific stimulation (9,25). Although these in vitro studies suggest physiologic functions for P2Y6 receptors in immune responses and inflammation, not all such functions have been demonstrated in vivo.
Given its distribution on cells of both the innate and adaptive immune systems, we suspected that P2Y6 receptors might play a role in the pathogenesis of allergic pulmonary inflammation. Here, we demonstrate that P2Y6 receptors are endogenous suppressors of T cell-driven pulmonary pathology induced by exposure to the allergens of the house dust mite Dermatophagoides farinae, which are potent sensitizers in asthma. Newly created C57BL/6 mice bearing a conditional deletion of the P2Y6 receptor (p2ry6 (flox/flox);cre/+ mice) displayed sharply increased pulmonary inflammation, lymph node hyperplasia, and allergen-induced Th1 and Th2 recall responses compared with P2Y6-sufficient littermate controls [p2ry6 (flox/flox);+/+; (+/+) mice] when exposed intranasally to an extract (Df) of Dermatophagoides farinae. P2Y6 receptors were constitutively expressed at low levels by alveolar and lung interstitial macrophages, but absent on splenic and parabronchial lymph node (PLN) T cells from naïve mice. However, P2Y6 receptor expression was induced on both CD4+ and CD8+ T cells from PLNs of +/+ mice exposed to Df. The absence of P2Y6 receptors from CD4+ T cells of Df-challenged mice, but not from lung-derived macrophages used as APCs, was sufficient to increase Th1 and Th2 cytokine generation in an antigen restimulation assay. Thus, the inducible expression of P2Y6 receptors by T cells permits endogenous UDP to modify cellular activation and cytokine production, dampening the inflammatory response to the clinically relevant allergens of house dust mites.
Materials and Methods
Mice
A 12.7 kb region of the p2ry6 gene was subcloned from a B6 bacterial artificial chromosome (BAC) clone using homologous recombination. A single loxP site was inserted at the 5′ side of exon 3 and a PGK-Neo cassette flanked by FRT sites and loxP sites was inserted at the 3′ of exon 3. The region of the targeting vector was designed such that the long (LA) and the short (SA) homology arms extended 9.6 kb at the 5′ side and 2.1 kb at the 3′ of the cassette, respectively. The structure of the targeting vector was confirmed by restriction analysis after each modification step and by sequencing using primers designed to read from the selection cassette into the 3′ of the LA (N7) and the 5′ end of the SA (N1) and primers annealing to the vector sequence (P6 and T7) and reading into the 5′ and 3′ ends of the BAC subclone. The BAC was subcloned into a 2.4 kb subclone vector containing an ampicillin cassette for retrotransformation of the construct prior to electroporation. The resultant targeting vector was linearized and electroporated into 129/Sv mouse embryonic stem (ES) cells and cells were positively selected with G418. Six verified ES clones were microinjected into blastocysts from C57BL/6 mice and chimeric mice were obtained and crossed with C57BL/6 mice to produce male and female p2ry6 (flox/+) mice. To generate p2ry6 (flox/flox) and p2ry6 (flox/flox); cre/+ mice on a C57BL/6 × 129 mixed background, p2ry6 (flox/+) mice were intracrossed to produce p2ry6 (flox/flox) mice. P2ry6 (flox/flox) mice were then mated with C57BL/6 mice homozygous for the Cre recombinase (cre/cre, C57BL/6 rosa26CreER(T2); Taconic-Artemis GmbH, Cologne, Germany) to obtain p2ry6 (flox/+);cre/+ mice that were then mated with p2ry6 (flox/flox) mice to produce the experimental mice, p2ry6 (flox/flox) (+/+ mice) and p2ry6 (flox/flox);cre/+ (Fig. 1B). Once p2ry6 (flox/flox) and p2ry6 (flox/flox); cre/+ were obtained, they were also used as breeders for the maintenance of the colony (Fig. 1). The Cre recombinase was induced in 5 – 11 week-old p2ry6 (flox/flox);cre/+ mice, by administering tamoxifen (1 mg; Sigma), dissolved in a mixture of ethanol/sunflower seed oil (1:4.5, v/v), by intraperitoneal injections on 5 consecutive days (29). Cohorts of sex- and age-matched p2ry6 (flox/flox) mice were also administered Tamoxifen simultaneously to ensure an equal treatment of the two strains. Tamoxifen-mediated induction of Cre recombinase in p2ry6 (flox/flox); cre/+ mice resulted in the disruption of p2yr6 gene and a loss of protein expression (Fig. 3). Mice were housed under pathogen-free conditions and maintained on a 12-hour light/dark cycle for the entire duration of the studies. All the studies described in this paper were approved by the Animal Care and Use Committee of the Dana Farber Cancer Institute (Boston, MA).
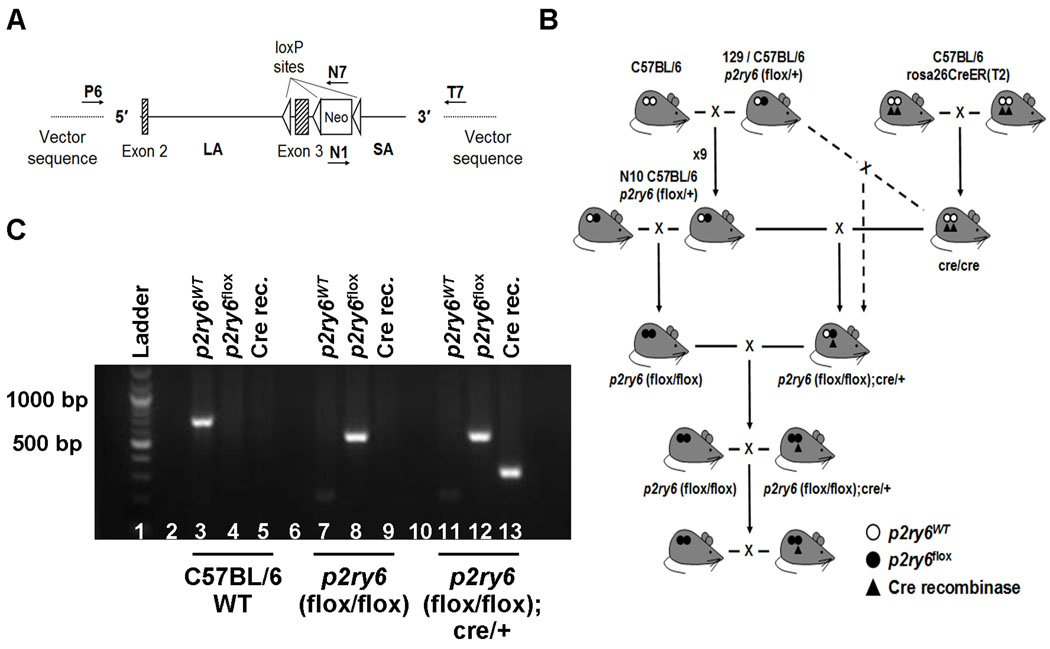
Figure 1. Generation of +/+ and p2ry6 (flox/flox);cre/+ mice.
(A) The conditional gene-targeting vector was obtained from an ~12.7 kb B6 bacterial artificial chromosome subclone and contained a long homology arm (LA; 9.6 kb), p2ry6 exons 2 and 3, the latter flanked at the 5′ side by a single loxP site and at the 3′ side by a PKG-neo cassette with loxP and FRT sites, and a short homology arm (SA; 2.1 kb). The target region was ~1.8 kb and included exon 3. The annealing sites of the primers used to confirm the structure of the vector are indicated as P6, N7, N1, and T7. (B) Schematic representation of the breeding protocol used to generate +/+ and p2ry6 (flox/flox);cre/+ mice. p2ry6 (flox/flox) mice on a C57BL/6 × 129 mixed background (dashed line) or on a C57BL/6 background (solid line) were initially mated with cre/cre mice to generate +/+ and p2ry6 (flox/flox); cre/+ strains. (C) PCR amplification products of the wild-type (p2ry6WT) and floxed (p2ry6flox) p2ry6 gene and of the Cre recombinase in C57BL/6 wild-type (lanes 3–5), p2ry6 (flox/flox) (lanes 7–9) and p2ry6 (flox/flox);cre/+ (lanes 11–13) mice, resolved on a 2% agarose gel.
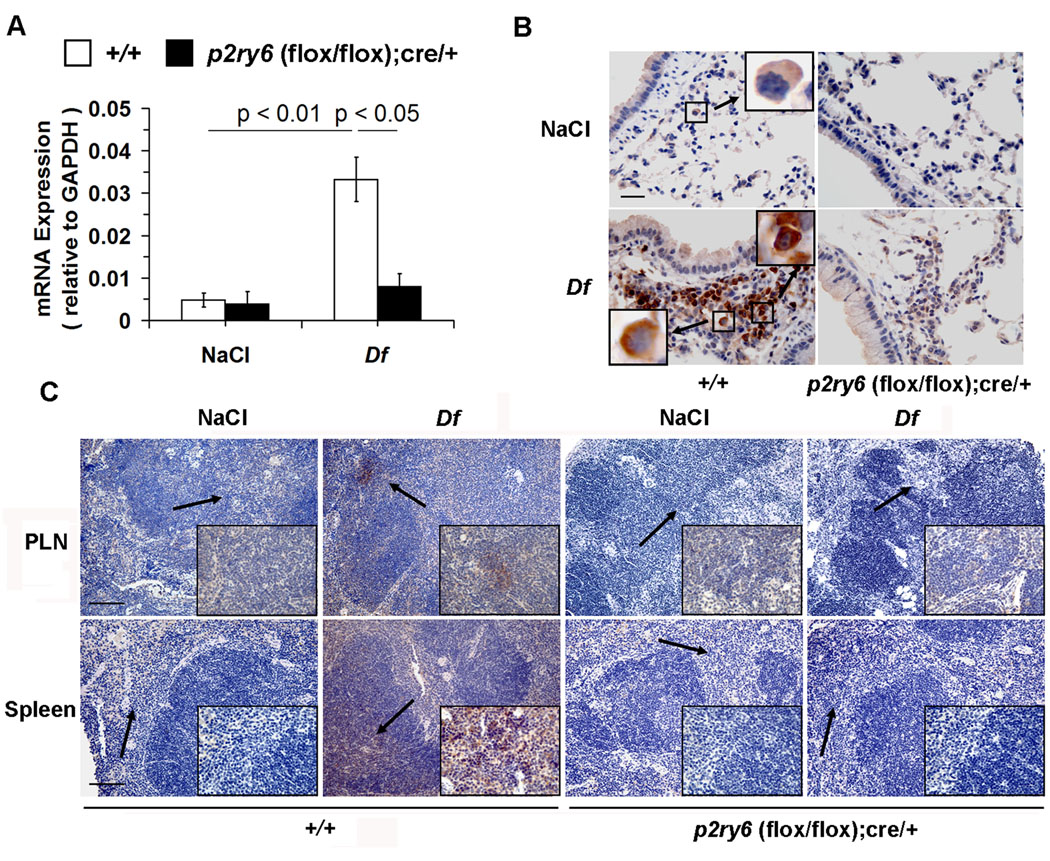
Figure 3. Expression of P2Y6 receptors in the lungs, PLNs and spleen.
(A) qPCR analysis of P2Y6 receptor mRNA in the lungs of NaCl- and Df-treated +/+ (open bars; n = 6 and 18, respectively) and p2ry6 (flox/flox); cre/+ (filled bars; n = 6 and 15) mice.
(B) Immunohistochemical analysis of P2Y6 receptors in the lungs from +/+ and p2ry6 (flox/flox); cre/+ mice exposed to NaCl and Df intranasally. P2Y6 receptor protein, indicated by the brown staining, was detected on cells with the morphology consistent with macrophages (upper left panel, insert) in the lung of +/+ naïve mice and on both macrophages (lower left panel, left insert) and lymphocytes (right insert) in the bronchus-associated lymphoid tissues of +/+ mice. No staining was detected in the lung of NaCl- and Df-treated p2ry6 (flox/flox); cre/+ mice (right panels). Scale bar, 25 µm. (C) Immunohistochemistry of P2Y6 receptors in PLNs and spleen of NaCl- and Df-treated +/+ and p2ry6 (flox/flox); cre/+ mice. P2Y6 receptors were detected on cells located in paracortical T cell-dependent areas of the organs (arrows), as shown in the inserts at higher magnification. Scale bars, 100 µm. Values in (A) are mean ± SEM from three independent experiments. Pictures in (B) and (C) are from one representative mouse per group from one of two independent experiments with similar results. Original magnification, x63 (B), x20 and x63 (C).
Induction of pulmonary inflammation
Ten days after the administration of tamoxifen, pulmonary inflammation was induced in age- (7–13 weeks) and sex-matched +/+ and p2ry6 (flox/flox);cre/+ mice by intranasal instillation of 3 µg of the Dermatophagoides farinae house dust mite extract (Df; Greer Laboratories, Lenoir, NC) in 20 µl of NaCl 0.9 % (containing <0.005 EU/ml of endotoxin; Sigma, St. Louis, MO) twice a week for 3 weeks, as described (30,31). In selected experiments the amount of Df was lowered to 1 µg/dose. Control groups of +/+ and p2ry6 (flox/flox); cre/+ mice received NaCl 0.9% alone. The same protocol was used for the experiments with the cre/+ mice. Twenty four h after the last treatment, mice were euthanized and blood was collected by cardiac puncture and centrifuged to obtain serum. Bronchoalveolar lavage (BAL) was performed with three aliquots of 0.7 ml of ice-cold PBS containing 10% FBS and 0.5 mM EDTA. BAL fluid cells were cytocentrifuged onto slides, stained with Diff-quick (Fisher Diagnostic, Middletown, VA), and differentially counted as mononuclear cells (MNCs; monocytes/macrophages and lymphocytes), neutrophils, or eosinophils, according to the standard hemocytological criteria.
Histological evaluation of pulmonary inflammation
Left lungs were harvested at the time of euthanasia, fixed and embedded in glycolmethacrylate or in paraffin (32). Two-µm -thick glycolmethacrylate sections were stained with hematoxylin-azue-2 eosin (H&E) or by the periodic acid-Schiff (PAS) reaction to depict mucus-secreting epithelial cells (goblet cells). The extent of cellular infiltration in the BVBs and the number of PAS-positive goblet cells were evaluated from each animal in the experimental groups by a pathologist blind to mouse strain and procedure.
Real-time quantitative PCR (qPCR) of mRNA transcripts in the lung and PLNs
Right lungs were collected at time of euthanasia and snap frozen. Freshly isolated PLN cells were pooled from 5–7 mice/group. Total RNA was isolated from tissue homogenates and from PLN cells with Tri-Reagent (Sigma), reverse transcribed into cDNA (RT2 First Strand kit, SABiosciences, Frederick, MD), and assayed by real-time qPCR for mouse P2Y6, IL-5, IL-13, the mucus-associated proteins Muc5ac and Clca3/Gob-5, the transcription factors T-bet, GATA-3, FoxP3, and ROR-γT, the chemokines CCL2, CCL11, and CXCL2, and GAPDH on a Mx3005P thermal cycler (Stratagene) with the use of SYBR®Green/ROX® master mix (SABiosciences). The ratio of each mRNA relative to the GAPDH mRNA was calculated with the ΔΔCt method. The primers used for mouse P2Y6 (16), IL-5, IL-13, Muc5ac, Clca3/Gob-5 and GAPDH were previously reported (31). For the transcription factors, the fold-induction was calculated based on the ratio of corrected expression in the samples from Df-treated mice and from saline-treated controls.
Immunohistochemical analysis of P2Y6 in lungs, PLNs and spleen
Sections of paraffin-embedded lungs, PLNs and spleens were deparaffinized and rehydrated. Antigen retrieval was performed with Target Retrieval Solution (Dako, Denmark) at 97°C for 30 min. After blocking with 10% chicken serum (Santa Cruz Biotechnologies, Santa Cruz, CA), sections were incubated (1 h, 37°C) with 43 µg/ml of a rabbit anti-P2Y6 Ab (20) and then with the peroxidase-conjugated rabbit ABC staining system (Santa Cruz) according to the manufacturer’s instructions. Slides were analyzed with a Leica DM LB2 microscope (Leica Microsystems, Wetzlar, Germany) and pictures captured with a Nikon digital camera DXM 1200 with Nikon ACT-1 (version 2.70) image acquisition software.
Flow cytometry analysis of PLN cells
Pooled PLNs from NaCl- and Df-treated +/+ and p2ry6 (flox/flox); cre/+ mice were homogenized and red cells were lysed. The cells obtained were fixed, permeabilized by incubation in FACS buffer [PBS buffer containing 0.5% bovine serum albumin (Sigma)] + 0.1% saponin (from Quillaja saponaria, Sigma) and blocked in FACS buffer containing 10% normal mouse serum and 1% anti-mouse CD16/CD32 (Mouse BD Fc Block™, BD Biosciences, San Jose, CA). Cells were washed and incubated (45 min, 4°C) with the following Abs: allophycocyanin-CD4 (clone RM4–5), allophycocyanin-CD8b (H35-17.2), PE-Cy7-CD11c (N418), PerCP-Cy5.5-B220 (RA3–6B2) (eBioscience), and Alexa Fluor 488-labeled (Zenon Alexa Fluor 488 rabbit IgG labeling kit, Invitrogen) rabbit anti-P2Y6 [10 µg/ml, directed against a conserved peptide within the second intracellular loop of the human P2Y6 receptor (20). The corresponding isotypes (eBioscience) or Alexa Fluor 488-labeled normal rabbit IgG (Jackson ImmunoResearch Laboratories) were used as controls. Annexin V staining was performed as described (33) on the CD4+ gate. The acquisition was performed on a FACSCanto flow cytometer with FACSDiva software (BD Biosciences), and data were analyzed with FlowJo (Tree Star, Ashland, OR).
In vitro restimulation of lymph node cells with Df
PLNs were collected from the upper-right chest of each mouse and homogenized in complete medium [RPMI, 10% FBS, 1% non-essential amino acids, 2 mM L-glutamine, 100 U/ml penicillin, 100 µg/ml streptomycin, 50 µM 2-ME (Sigma)], plus 25 mM HEPES, and 1 mM sodium pyruvate. The red cells were lysed, and 4 × 106 nucleated cells were incubated for 72 h with medium alone or containing 20 µg/ml Df (30). The concentrations of IL-2, IL-4, IL-5, IL-10, IL-13, IL-17A, and IFN-γ released in the supernatants were measured by ELISA (eBioscience, San Diego, CA). The amount of cytokine per 106 PLN cells and the total number of PLN cells were used to calculate the total amount of cytokine generated per lymph node.
For the ELISPOT analysis, 2.5 × 105 PLN nucleated cells were incubated in medium containing Df (20 µg/ml) for 72 h on 96-well cell culture plates with PVDF filter membranes (Millipore, Billerica, MA), pre-coated with Abs against IL-4, IL-5, IL-13, IL-17A, and IFN-γ (eBioscience). The spots were detected with an ELISPOT kit (eBioscience) according to the manufacturer’s instructions and counted with a CTL Immunospot® Analyzer (Cellular Technology, Shaker Heights, OH). For each cytokine the total number of producing cells was calculated by multiplying the frequency of producing cells by the total number of PLN cells.
PLN CD4+ T cell proliferation
PLN cells from NaCl- or Df-treated +/+ and p2ry6 (flox/flox);cre/+ mice were obtained, incubated (10 min, 37°C) in PBS with 1 µM CFDA-SE (eBioscience) and extensively washed in culture medium. 4 × 106 CFDA-SE-labeled cells were then added to 24-well plates pre-coated with an anti-CD3 Ab (1 µg/ml overnight) and cultured in the presence of soluble anti-CD28 Ab (1 µg/ml) for 72 h. At the end of the incubation cells were harvested, washed in FACS buffer, blocked and incubated (45 min, 4°C) with APC-CD4 Ab (RM4–5, eBioscience). Data was acquired on a FACSCanto flow cytometer with FACSDiva software. The CFDA-SE profile was evaluated on the CD4+ gate and analyzed with FlowJo. The cells falling into the lowest CFSE peak were counted as “proliferating”.
Isolation of lung macrophages and T cells for confocal microscopy
Lung macrophages were obtained using a modification of previous protocols (34,35). After removing airway cells and alveolar macrophages by extensive BAL with PBS containing EDTA, lungs were homogenized through a 70 µm mesh in ice cold complete medium, washed and incubated (30 min, 37°C) with 428 U/ml Collagenase IV (Worthington, Lakewood, NJ) and 20 µg/ml DNAse I (Roche, Mannheim, Germany). The obtained cell suspension was washed and layered onto a Percoll® gradient (60% and 40%). After centrifugation the cells at each interface were collected, washed, and centrifuged again on a second density gradient. At the end of the procedure the obtained cells, representing an enriched (>85%) population of interstitial macrophages, were stained with Diff-quick (Fisher Diagnostic) or analyzed by flow cytometry with the following Abs: allophycocianin-CD68 (clone FA-11; AbD Serotec), FITC-MHC-II (M5/114.15.2), PE-Cy7-CD11c, (N418), PE-Cy7-CD3e (145-2C11), FITC-CD4 (RM4–5), PE-Cy7-CD8a (53-6.7), and PerCP-Cy5.5-B220 (RA3-6B2) (eBioscience). CD4+ cells were purified from pooled lungs of 5 mice/group using a FACSAria II high-speed cell sorter (Dana-Farber Cancer Institute Flow Cytometry Core). For confocal microscopy analysis, the macrophages were further enriched by adherence (overnight, 37°C) on cover slips. After removing non-adherent cells by washing, adherent macrophages were fixed with 2% paraformaldehyde (Sigma) and permeabilized with PBS containing 0.1% saponin (36). Cells were stained with goat polyclonal anti-P2Y6 Ab or goat IgG as negative control (10 µg/ml; Santa Cruz), and counterstained with FITC-conjugated donkey anti-rabbit IgG (Jackson ImmunoResearch). Z-stack images were acquired with a Nikon C1 plus laser scanner confocal system combined with an Eclipse TE2000-U inverted microscope with a x60 oil PlanApo NA 1.4 objective lens (36).
Co-cultures of lung macrophages and splenic CD4+ T cells
To purify CD4+ T cells, spleens of NaCl- or Df-treated +/+ and p2ry6 (flox/flox); cre/+ mice were homogenized and nucleated cells were blocked and incubated (45 min, 4°C) with PE-Cy7-CD3e (clone 145-2C11) and FITC-CD4 (RM4–5) Abs (eBioscience). The double positive CD3+/CD4+ cells were sorted by flow cytometry (Dana Farber Cancer Institute Flow Cytometry Facility, Boston, MA). Lung interstitial macrophages (1 × 105/ml) and CD3+/CD4+ cells (7.5 × 105/ml) were cocultured in either medium alone or containing 20 µg/ml Df for 72 h. At the end of the incubation the concentrations of IL-5, IL-13, and IFN-γ released in the supernatants were measured by ELISA (eBioscience).
Statistics
Non-parametric Mann-Whitney test and Kruskal-Wallis test with Dunn`s post test correction for multiple comparisons were used to compare two and three or more groups, respectively. Analysis was performed with Prism software (GraphPad, La Jolla, CA). Values of p < 0.05 were considered significant.
Results
Deletion of P2Y6 receptors increases pulmonary inflammation induced by Df allergen
The p2ry6 allele was deleted by flanking the gene with P-lox sites (Fig. 1A). The p2ry6 (flox/flox); cre/+ mice and their +/+ littermate controls (Fig. 1B) were treated with tamoxifen for 5 consecutive days intraperitoneally. PCR analysis confirmed the genotype of the mice used in the experiments (Fig. 1C). Immunohistochemistry confirmed the absence of the P2Y6 receptor protein in the lungs, PLN and spleen of the p2ry6 (flox/flox); cre/+ mice after tamoxifen treatment (Fig. 3). The initial experiments were performed with F1-F5 C57BL/6 × 129 mice and littermate controls. The results obtained in these experiments were similar to those obtained after backcrossing the mice for 10 generations to the C57BL/6 background (Fig. 2A).
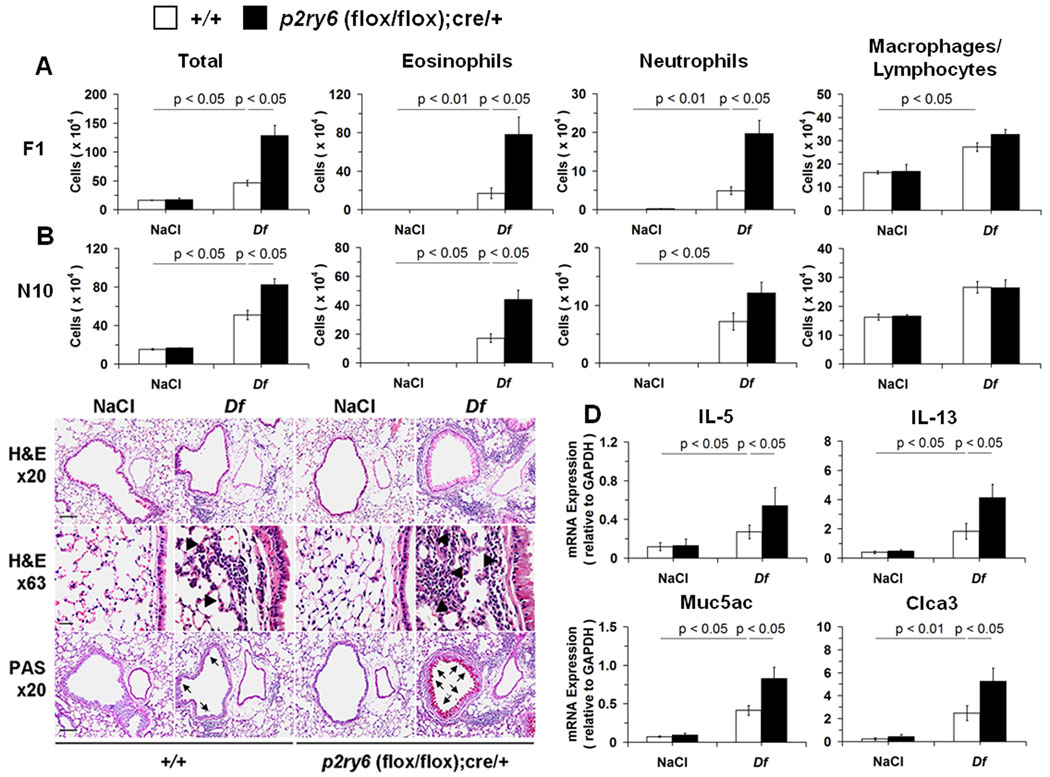
Figure 2. Df-induced pulmonary inflammation in NaCl- and Df-treated +/+ and p2ry6 (flox/flox); cre/+ mice.
(A) Total and differential cell counts from BAL fluid of NaCl-and Df-treated +/+ (open bars; n = 9 and 23, respectively) and p2ry6 (flox/flox); cre/+ (filled bars; n = 9 and 18) mice at the F1 stage of breeding. (B) Total and differential cell counts from BAL fluid of NaCl-and Df-treated C57BL/6 +/+ (open bars; n = 5 and 19, respectively) and p2ry6 (flox/flox); cre/+ (filled bars; n = 5 and 20) mice at the N10 stage. (C) Tissue sections of lung showing BVBs from NaCl- and Df-treated +/+ and p2ry6 (flox/flox);cre/+ mice were stained with hematoxylin-azue-2 eosin for assessing inflammatory cell infiltrates (H&E x20) and demonstrating tissue eosinophils (H&E x63, arrow heads) or by PAS reaction for depicting mucus-secreting cells (PAS x20, arrows). Scale bars, 100 (x20) or 25 (x63) µm. (D) Expression of mRNA encoding cytokines and mucus-associated proteins in the lung tissue of NaCl- and Df-treated +/+ (n = 6 and 18, respectively) and p2ry6 (flox/flox); cre/+ (n = 6 and 15) mice, measured by qPCR. Data are expressed as ratio of the indicated mRNA expression relative to GAPDH. Values are mean ± SEM from four (A) and three (B) independent experiments. Pictures in (C) are from one representative mouse per group from one of three independent experiments with similar results. Original magnifications, x20 and x63.
To determine whether P2Y6 receptors played a role in the development of allergic pulmonary inflammation, DF (3 µg of protein) or saline was administered intranasally, twice weekly for three consecutive weeks (25) to cohorts of +/+ and p2ry6 (flox/flox); cre/+ mice after both genotypes were treated with tamoxifen. DF caused increased BAL fluid cell numbers, eosinophils, neutrophils, macrophages, and lymphocytes. However, the total number of cells in BAL fluid of the Df-treated p2ry6 (flox/flox); cre/+ mice exceed that of the +/+ controls by more than 2-fold (Fig. 2A). Compared to the Df-treated +/+ controls, the Df-treated p2ry6 (flox/flox); cre/+ mice showed ~5-fold higher numbers of BAL fluid eosinophils and neutrophils (Fig. 2A). Similar results were obtained after backcrossing the initial F1 mice to the C57BL/6 background for 10 generations (Fig. 2B). Tamoxifen-treated Cre/+ mice without the fluxed p2ry6 allele showed identical degrees of Df-induced inflammation, PLN cell counts, and PLN cytokine production to WT controls (Supplemental Fig. 1A, 1B, and 1C, respectively), indicating that activation of Cre recombinase per se did not alter the phenotype.
Because the peribronchial inflammation caused by Df at 3 µg was too severe to show substantial differences between the genotypes (data not shown), we reduced the dose of Df to 1 µg in selected experiments to determine whether the deletion of P2Y6 receptors altered pulmonary pathology at a lower dose of antigen. At this allergen dose, the bronchovascular bundles (BVBs) of the p2ry6 (flox/flox);cre/+ mice contained more extensive inflammatory infiltrates consisting of mononuclear cells along with eosinophils, neutrophils, lymphocytes and plasma cells than did the +/+ controls (Fig. 2C). In addition, the bronchial epithelium of the p2ry6 (flox/flox);cre/+ mice demonstrated more goblet cell metaplasia than did the +/+ controls (Fig. 2C). Higher levels of expression of IL-5 and IL-13 mRNA transcripts were present in the lungs of the Df-treated p2ry6 (flox/flox);cre/+ mice than in the lungs of the +/+ mice, with similar trends for the goblet cell-associated transcripts Muc5ac and Clca3/Gob-5 (Fig. 2D). There were no significant differences in the levels of transcripts encoding CCL2 (a monocyte chemoattractant), CCL11 (an eosinophil chemoattractant) or CXCL1 (a neutrophil chemoattractant) (Supplemental Fig. 2) between the lungs of the genotypes.
Cellular distribution of P2Y6 receptors in the lungs and lymphoid tissues
Because the deletion of P2Y6 receptors resulted in increased pulmonary inflammation in response to Df, we sought to determine the cellular distribution of P2Y6 receptors in the lungs and associated secondary lymphoid organs of +/+ mice, and whether this distribution and the level of receptor expression changed with inflammation. qPCR revealed low constitutive levels of P2Y6 receptor mRNA in the lungs of naïve +/+ mice. These levels sharply increased in response to intranasal Df (Fig. 3A). Immunohistochemistry using a rabbit polyclonal anti-P2Y6 Ab (20) revealed that P2Y6 receptor protein localized predominantly to cells with the morphologic appearance of macrophages in the alveolar and interstitial spaces in saline-treated +/+ mice (Fig. 3B). Challenge of +/+ mice with Df induced the accumulation of mononuclear cells in the bronchus-associated lymphoid tissue of the BVBs, which stained strongly for P2Y6 receptor protein. Based on cell size and morphology, these cells appeared to be a mixture of lymphocytes and monocyte-like cells (Fig. 3B). There was negligible staining of the resident epithelial, endothelial, and smooth muscle cells, and no significant staining of the infiltrating granulocytes.
Immunostaining of the spleen and PLNs from naïve +/+ mice revealed negligible staining for P2Y6 receptor protein. In contrast, distinct populations of P2Y6 receptor-positive cells appeared in the paracortical zones of the spleen and PLNs from Df-treated +/+ mice (Fig. 3C), but not in the germinal centers. Neither the lung (Fig. 3B) nor the lymphoid tissues (Fig. 3C) of the saline- or Df-treated p2ry6 (flox/flox);cre/+ mice exhibited staining for P2Y6 receptor protein.
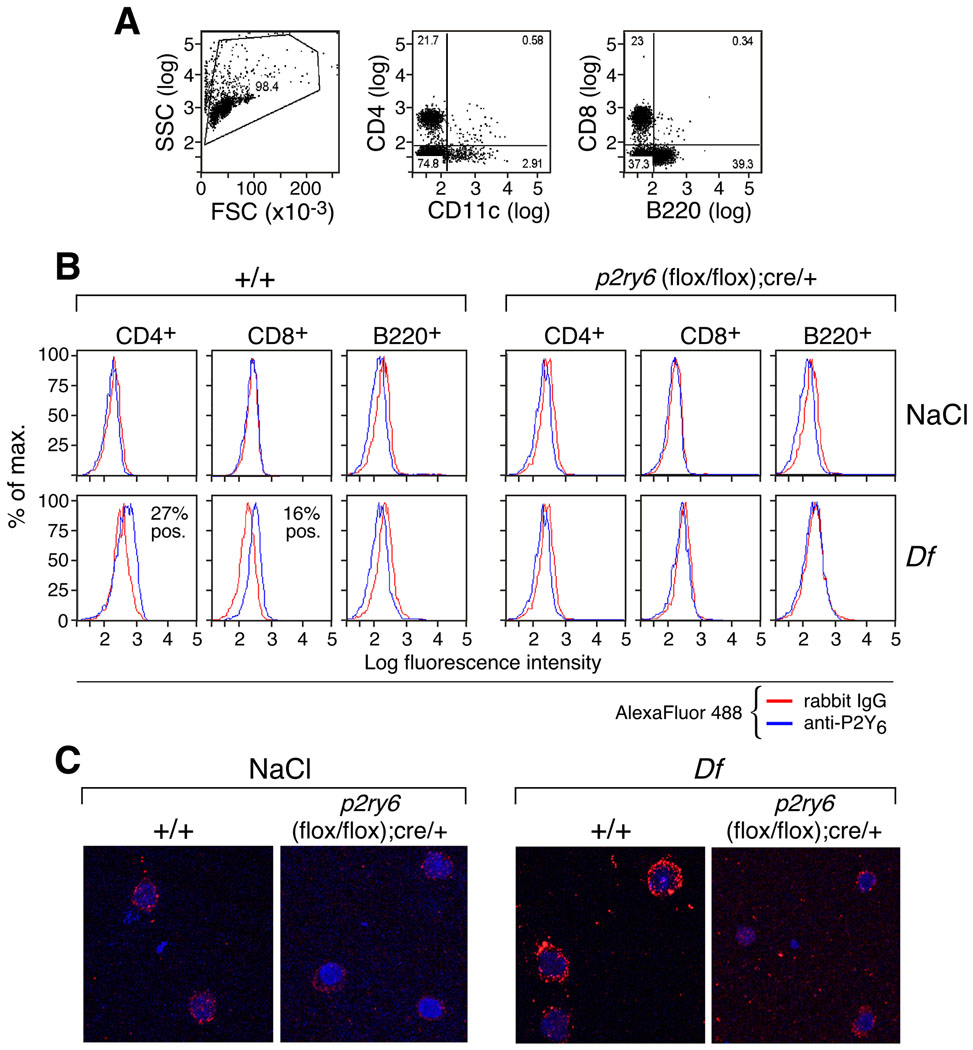
To identify the lymphoid cells expressing P2Y6 receptors after treatment with Df, we performed flow cytometry using the rabbit anti-P2Y6 Ab on the dispersed spleen cells from the +/+ mice. Abs against CD4, CD8, B220 (CD45R), and CD11c were used to identify helper and cytotoxic T cells, B cells and APCs, respectively (Fig. 4A). No P2Y6-expressing lymphoid cells were identified in the spleens from the naïve +/+ mice (Fig. 4B). In contrast, both CD4+ and (to a lesser extent) CD8+ splenic T cells from the Df-treated +/+ mice showed modest staining for P2Y6 (Fig. 4B). B220+ B cells did not express P2Y6 receptors, whether derived from naïve or Df-treated +/+ mice. There were too few CD11c+ cells to analyze, and no staining for P2Y6 receptors was observed on any cell populations in the spleens from saline- or Df-treated p2ry6 (flox/flox);cre/+ mice (Fig. 4B). The percentages of CD4+, CD8+, and B220+ cells in the lymph nodes did not differ between the two strains (not shown). Confocal imaging of CD4+ cells purified from the dispersed lungs of Df-treated wild-type mice confirmed prominent P2Y6 receptor staining, which was not observed in lung T cells from saline-treated +/+ mice (Fig. 4C).
Figure 4. Cytofluorographic detection of P2Y6 receptors on PLN cell subsets.
(A) Dot plots showing the FSC/SSC gated cells and the analysis of the expression of CD4, CD11c, CD8, and B220 on PLN cells from Df-treated +/+ mice. Percentages of positive and negative cells are displayed. (B) Histograms showing the expression of P2Y6 receptors (blue lines) vs. isotype control (red lines) on CD4 +, CD8+, and B220+-gated PLN cells in NaCl- and Df-treated +/+ and p2ry6 (flox/flox);cre/+ mice. Percent positives are displayed. (C) Confocal images of CD4+ cells purified from the lungs of the indicated NaCl- or Df-challenged strains. Panels are from one experiment representative of two with similar results.
Effects of P2Y6 receptors on the adaptive immune response to Df
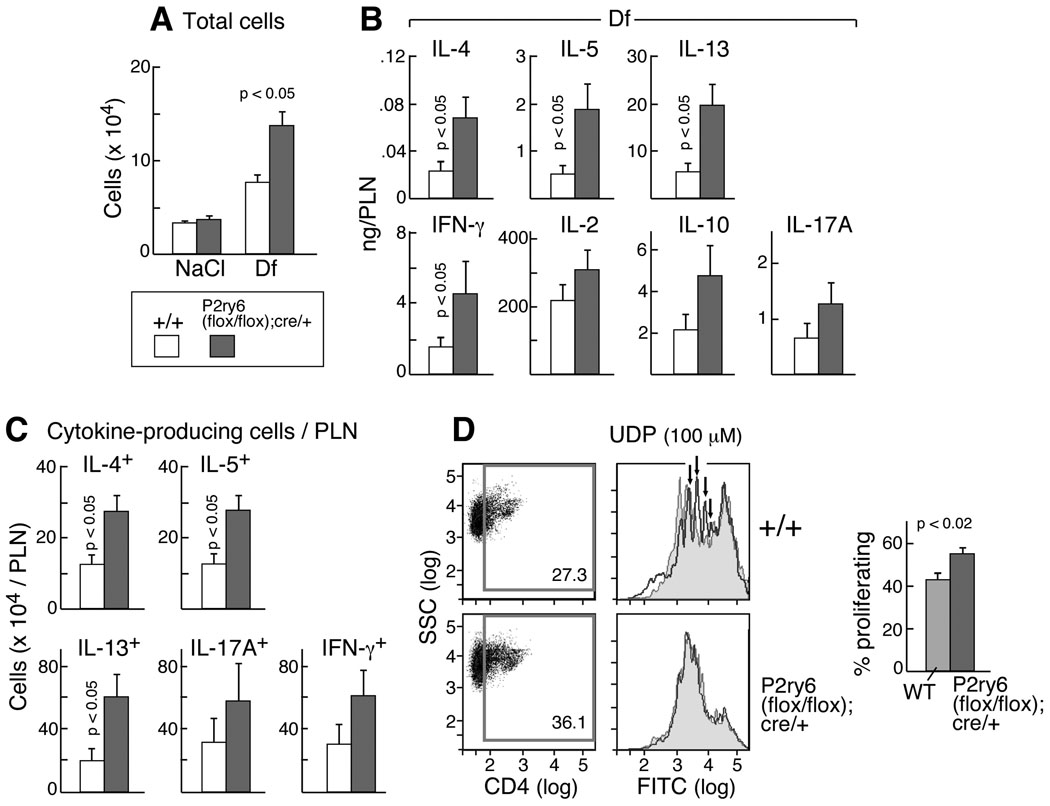
To determine the role of P2Y6 receptors on control of recall responses to the Df antigens in sensitized mice, we evaluated the production of proinflammatory and immunoregulatory cytokines from restimulated PLN cells from the Df-treated p2ry6 (flox/flox);cre/+ mice and +/+ controls. The PLNs from the Df-treated p2ry6 (flox/flox);cre/+ mice were consistently larger than those from the +/+ controls, and contained more total cells (Fig. 5A). Equal numbers of PLN cells isolated from NaCl- and Df-treated +/+ and p2ry6 (flox/flox);cre/+ mice were cultured in vitro in either medium alone or containing Df. At the end of the culture, the concentrations of IL-2, IL-4, IL-5, IL-10, IL-13, IL-17A, and IFN-γ released in the supernatants were evaluated by ELISA. Restimulated PLN cells from Df-treated p2ry6 (flox/flox);cre/+ mice generated more IL-4, IL-5, IL-13, and IFN-γ than did cells from +/+ mice (Fig. 5B). The amounts of IL-2, IL-10 and IL-17A also tended to be higher but did not reach significance (P = 0.26, 0.15 and 0.12, respectively). The results obtained from the PLN cells from the N10 mice were similar (Supplemental Figure. 3), and restimulation of splenocytes yielded results similar to the PLN cells (not shown). To determine whether the increased quantities of cytokines reflected increased numbers of cytokine-generating cells in the PLNs from Df-treated p2ry6 (flox/flox); cre/+ mice, we performed ELISPOT assays. Consistent with the ELISA data, the PLNs of Df-treated p2ry6 (flox/flox); cre/+ mice contained increased numbers of cells expressing IL-4, IL-5, and IL-13 protein than did the PLNs of the Df-treated +/+ controls (Fig. 5C). The differences in IFN-γ and IL-17A were not significant (p = 0.07 and 0.10, respectively). Quantitative PCR analysis of the Df-treated PLN cells showed no differences between strains in the induced expressions of the Th1-associated transcription factor T-bet, the Th2-associated transcription factor GATA-3, (the regulatory T cell (Treg) associated transcription factor FoxP3, and the Th17 transcription factor ROR-γt (Supplemental Fig. 4).
Figure 5. PLN cellularity and T cell responses.
(A) PLN cell numbers from NaCl- and Df-treated +/+ (open bars; n = 5 and 17, respectively) and p2ry6 (flox/flox);cre/+ (filled bars; n = 5 and 17) mice. (B) Cytokine release from restimulated PLN cells of Df-treated +/+ (n = 16) and p2ry6 (flox/flox); cre/+ (n = 17) mice. (C) Number of cytokine-producing cells in PLN cells of Df-treated +/+ (n = 15) and p2ry6 (flox/flox); cre/+ (n = 13) mice. Values in (A), (B), and (C) are mean ± SEM from three independent experiments. (D) Proliferation (based on CFDA-SE dilution) of CD4+-gated PLN cells from Df-treated +/+ and p2ry6 (flox/flox); cre/+ mice (N10 generation) cultured with plate-bound anti-CD3 and soluble anti-CD28 Abs. The mean ± SEM percent of proliferating cells from 5 mice/group are shown (right). Histograms showing the CFSE dilution of the CD4+ gate of polyclonally stimulated spleen cells treated with UDP (100 µM, blue) are overplayed with histograms of untreated cells (red) from the same strains in one experiment representative of two performed.
To determine whether the absence of P2Y6 receptors changed the rate of helper T cell proliferation, PLN cells were loaded with CFDA-SE before restimulation and the dilution of the dye was measured cytofluorographically as an index of proliferation. Since the CFDA-SE dilution signal in cultures of PLN cells restimulated with Df was below the limit of detection (data not shown), we performed the assay by using a T cell polyclonal stimulation with anti-CD3 and anti-CD28 Abs. Percentages of polyclonally stimulated proliferating CD4+ T cells were modestly but significantly higher in the PLNs of p2ry6 (flox/flox);cre/+ mice than in the PLNs of +/+ controls (Fig. 5D). Splenic CD4+ cells p2ry6 (flox/flox);cre/+ mice also displayed higher rates of proliferation in response to polyclonal stimulation than did +/+ control cells, and unlike the +/+ cells, exogenous UDP failed to suppress their proliferation (Fig. 5D). Percentages of apoptotic CD4 cells in the PLNs of the Df-treated p2ry6 (flox/flox);cre/+ mice were lower than in the PLNs from the +/+ controls (Supplemental Fig. 5). The p2ry6 (flox/flox); cre/+ mice exposed to Df showed higher levels of serum total IgE (Supplemental Fig. 6A), but they had comparable levels of Df-IgG1 (Supplemental Fig. 6B).
The absence of P2Y6 receptors on CD4+ T cells controls the recall response to Df-associated allergens
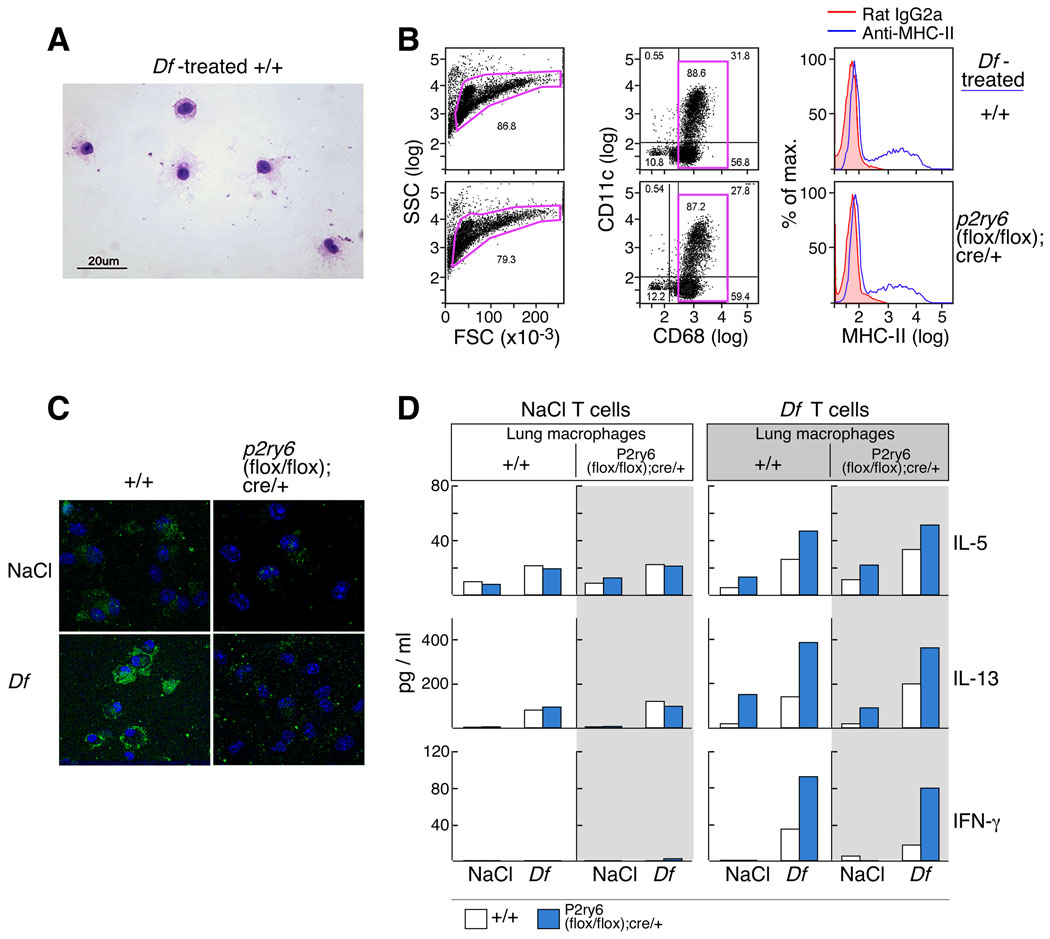
To determine whether the increased cytokine production from the restimulated PLNs reflected a direct suppressive effect of P2Y6 receptors expressed by T cells, or an effect of P2Y6 receptor-dependent modulation of APC functions, we developed a co-culture assay. Because mice with transgenic expression of Df-specific T cell receptor are not yet available, we purified CD3+/CD4+ T cells from the spleens of NaCl- and Df-treated +/+ and p2ry6 (flox/flox);cre/+ mice and incubated them with enriched interstitial macrophages from the lungs of each genotype and treatment as a source of APCs. Nearly all of these cells had morphologic features of macrophages (Fig. 6A Fig.), with >85% expressing the pan-macrophage marker CD68 (Fig. 6B) (37). Fewer than 5% of the cells expressed T cell markers (CD3, CD4 and CD8); while B220 was expressed by ~15% of the CD68+ cells (data not shown). The proportions of the cells expressing the APC markers CD11c and MHC-II (~30% for each marker) did not differ between genotypes (Fig. 6B), and confocal imaging confirmed that the macrophages from the Df-treated +/+ mice exhibited substantially increased expression of P2Y6 protein relative to the saline-treated controls (Fig. 6C). After 72 h of co-culture in the presence of Df, CD3+/CD4+ T cells from naïve mice of both genotypes generated small and comparable amounts of IL-5, IL-13 (Fig. 6, left panels). In contrast, CD3+/CD4+ T cells from Df-treated p2ry6 (flox/flox); cre/+ mice released more IL-5, IL-13, and IFN-γ than the cells from +/+mice in all three experiments performed (as shown for one experiment, Fig. 6D, right panels). There was no effect of the macrophage genotype; although macrophages from lungs of Df-treated mice supported higher levels of cytokine generation by CD4+ cells than did macrophages from the lungs of saline-treated controls (Fig. 6D).
Figure 6. Cytokine release from restimulated cocultures of CD4+ T cells and lung macrophages.
(A) Wrights and Giemsa stain showing enriched lung macrophages from Df-treated +/+ mice. (B) Forward and side scatter characteristics (left), CD68/CD11c staining (middle) and MHC-II staining (right) of the enriched macrophages from the indicated strains used in the co-culture assays. (D) Cytokine release from restimulated cocultures of CD4+ T cells and lung macrophages. Lung macrophages from NaCl- and Df-treated +/+ (unshaded) and p2ry6 (flox/flox); cre/+ (shaded) mice were co-cultured with CD4+ T cells from +/+ (open bars) and p2ry6 (flox/flox); cre/+ (filled bars) mice treated with NaCl (left panels) or Df (right panels). The results are from one experiment, which was repeated twice more with similar trends but with different magnitude of responses.
Discussion
This study uncovers a prominent role for the UDP-selective P2Y6 receptor in the control of pulmonary inflammation and cytokine generation in a model of allergic pulmonary inflammation. In our model, uracil nucleotides act at P2Y6 receptors that are inducibly expressed by T cells to suppress the development of Df antigen-responsive T cells and production of pathogenetic cytokines. In vitro studies indicate that UDP (acting presumably at P2Y6 receptors) amplifies effector cytokine production by epithelium (21), endothelium (28), macrophages (27), and mast cells (20). Although we cannot exclude important contributions from P2Y6 receptors on these other cell types in our model, our study suggests that upregulation of P2Y6 receptors on T cells and limiting T cell activation may be a dominant mechanism that limits the pathologic consequences of a pulmonary immune response, at least in response to a complex natural allergen.
Given the fact that multiple P2Y6 receptor-expressing cell types can contribute to the pathophysiology of the immune and inflammatory response to natural allergens, we tested the effect of P2Y6 receptor deletion in a model of intranasal Df challenge. Df contains glycans, proteases, and other stimuli that activate lung APCs and initiate a pathogenetic T cell response with the generation of Th1, Th2, and Th17 cytokines and the development of characteristic pulmonary inflammation (38,39). We generated a mouse with homozygous floxed p2ry6 alleles (Fig.1A) that were disrupted by tamoxifen treatment after introducing a single copy of a tamoxifen-sensitive Cre recombinase allele [p2ry6 (flox/flox);cre/+] (Figures 1B and 1C). These mice were fertile, healthy, and indistinguishable from littermate +/+ controls, similar to a previously reported conditional p2ry6−/− mice (16). Based on the role for P2Y6 receptors in amplifying cytokine production by immune cells in vitro, we anticipated that the deletion of this receptor might reduce inflammation in our model. We were surprised to find that the p2ry6 (flox/flox);cre/+ mice exhibited increased inflammation after Df administration compared with the +/+ littermates, showing significantly increased total cell counts, increased eosinophil and neutrophil counts in the BAL fluid (Figures 2A and 2B), as well as increased bronchovascular cellular infiltration and goblet cell metaplasia on histological analysis (Fig.2C). The increases in goblet cell-associated transcripts in the lungs of p2ry6 (flox/flox);cre/+ mice, which are characteristically controlled by the Th2 cytokine IL-13 (40–42), was paralleled by increased quantities of transcripts encoding IL-13, as well as IL-5, the major cytokine responsible for eosinophilia (43,44) (Fig.2D). Thus, the absence of P2Y6 receptors increased the expression of pathogenetic Th2 cytokines in the target tissue, leading to characteristic lung pathology.
While cytokine generation in models of allergen-induced pulmonary disease requires T cell activation following the presentation of antigens by APCs (39), several additional cell types, including epithelial cells, modify cytokine generation by recognizing non-protein components of dust mites (45–47). We determined the distribution of P2Y6 receptor expression in resident lung cells in both naïve and Df-treated mice. P2Y6 receptor staining in the lungs of naïve +/+ mice was restricted primarily to macrophages in both the alveolar and interstitial spaces of the lung (Fig.3B). The sharply increased expression of P2Y6 receptor transcript in the lungs of Df-treated +/+ mice (Fig.3A) was paralleled by the appearance of monocytoid and lymphocytoid cells that strongly expressed P2Y6 receptor protein, and that localized to the bronchus-associated lymphoid tissues (Fig.3B). Additionally, staining of the regional lymph nodes and of the spleen suggested that Df treatment of +/+ mice induced a P2Y6 receptor-expressing T cell population (Fig. 3C), and flow cytometry verified that PLN CD4+ cells (and, to a lesser extent, CD8+ cells) expressed P2Y6 receptors after exposure in vivo to allergen (Fig. 4, a pattern recapitulated by lung T cells (Fig. 4). The absence of P2Y6 receptor expression on B220+ B cells is consistent with the lack of immunostaining of germinal centers (Fig. 3C). Because there were very few CD11c+ APCs in the PLNs, we were unable to determine whether they expressed P2Y6 receptors, although macrophages enriched from the lung did express P2Y6 receptors that were upregulated with Df treatment (Fig. 6C).
We next sought to determine the effects of P2Y6 receptor deletion on the recall response to Df. The PLNs from the Df-treated p2ry6 (flox/flox);cre/+ mice contained more total cells (Fig. 5A) than did the PLNs from the +/+ controls. Although the total percentages of T cells in the PLNs did not differ between the strains (data not shown), the restimulated PLN cells from the Df-treated p2ry6 (flox/flox);cre/+ mice generated significantly higher amounts of Th2 and Th1 cytokines than did samples from the +/+ mice (Fig. 5B), paralleled by higher numbers of cytokine producing T cells (Fig. 5C). Thus, the effect of P2Y6 receptors on the control of antigen recall responses is not restricted to one lineage of effector T cell in this model. Moreover, the robust generation of IL-10 (Fig. 5B) by the samples from p2ry6 (flox/flox);cre/+ mice, and the trivial induction of mRNA encoding Foxp3 in PLN cells from +/+ and p2ry6 (flox/flox);cre/+ mice following exposure to Df (Supplemental Fig. 4) suggest that the absence of P2Y6 receptors does not selectively compromise regulatory T cell development as a mechanism for the phenotype observed. Thus, P2Y6 receptors induced by antigen stimulation play a homeostatic role in the control of memory T cell activation during recall responses to Df antigens. Although the low frequencies of Df-responsive T cells (estimated at ~1 in 100 lymph node cells) precluded detection of proliferation in a recall response to Df, polyclonally stimulated CD4+ PLN T cells from p2ry6 (flox/flox);cre/+ mice proliferated significantly faster than did +/+ control cells. The fact that exogenous UDP suppressed proliferation only in the CD4+ cells from +/+ controls (Fig. 5D) validates that P2Y6 receptors account entirely for the UDP response of T cells in this model. It is possible that both enhanced proliferation and attenuated apoptosis contribute to the relative PLN hyperplasia of the p2ry6 (flox/flox);cre/+ mice in this model. Proof of a homeostatic role for P2Y6 in the control of antigen-induced proliferation awaits the creation of transgenic mice with T cell receptors specific for Df antigens.
In addition to intrinsic properties of the responder T cells, APC-specific factors strongly influence the magnitude and nature of antigen-induced recall responses (39). To test whether the outcome of the Df-induced recall response was influenced more strongly by P2Y6 receptors expressed by APCs or by effector T cells, we purified P2Y6 receptor-deficient or -sufficient splenic CD4+ T cells and co-cultured these with either P2Y6 receptor-deficient or -sufficient lung macrophages as a source of APCs (Fig. 6) (48). The induction of inflammation by Df causes APCs to mature and become more effective in processing and presenting antigen, likely explaining the ability of the CD4+ cells to produce higher level of cytokines when co-cultured with APCs derived from Df-treated mice. The absence of P2Y6 receptors did not affect the expression of activated APC markers CD11c and MHC-II (Fig. 6). Importantly, the macrophage genotype had no influence on the levels of cytokines generated by responder T cells. Although the absolute quantities of cytokines produced varied in the three experiments performed, CD4+ cells from the Df-treated p2ry6 (flox/flox);cre/+ mice generated more IL-5, IL-13, and IFN-γ than did the CD4+ T cells from the +/+ controls in every experiment (as shown for one experiment, Fig. 6D). Thus, at least at the effector phase of the Df-induced immune response, the absence of P2Y6 on Df-treated CD4+ cells is both necessary and sufficient to amplify the production of cytokines in response to stimulation with the allergen in a recall response.
Our study demonstrates the first homeostatic role of P2Y6 receptors in the intrinsic control of effector T cell responses to clinically relevant allergens. The P2Y6 receptor mediates a suppressive function for the uracil nucleotides that are generated in the inflammatory milieu during exposure to dust mite allergens. Two recent studies reported that uridine, the precursor of UDP and UTP, has a protective function in two models of acute lung inflammation by acting on epithelial cells (49,50). Thus, uracil and its derivatives may suppress allergic pulmonary inflammation by several mechanisms, contrasting with the proinflammatory effects of adenine nucleotides in a similar context. Given the multitude of P2Y receptors that are expressed by cells of the immune system, it is critical to understand both the pro- and anti-inflammatory effects of any P2Y receptor that is envisioned as a potential target for drug development in the treatment of inflammatory diseases.
Supplementary Material
Acknowledgements
The authors wish to acknowledge Chunli Feng for outstanding technical support.
Footnotes
This work was funded by National Institute of Health Grants AI064226 (to B.B.), AI52353, AI31599, AI07306, and HL36110 and by generous contributions from the Vinik Family (to J.A.B). G.G. is supported in part by an educational grant from the Ph.D. Program in Clinical Pathophysiology and Experimental Medicine of the University of Naples “Federico II”, Naples, Italy.
Non-standard abbreviations used in this manuscript
BVB, bronchovascular bundle; Df, extract from Dermatophagoides farinae; GPCR, G protein-coupled receptor; MC, mast cell; P2Y, purinergic GPCR; PLN, parabronchial lymph node
The authors declare no conflict of interest.
References
- 1.Abbracchio MP, Burnstock G. Purinergic signalling: pathophysiological roles. Jpn. J. Pharmacol. 1998;78:113–145. doi: 10.1254/jjp.78.113. [DOI] [PubMed] [Google Scholar]
- 2.Burnstock G. Pathophysiology and therapeutic potential of purinergic signaling. Pharmacol. Rev. 2006;58:58–86. doi: 10.1124/pr.58.1.5. [DOI] [PubMed] [Google Scholar]
- 3.Bergfeld GR, Forrester T. Release of ATP from human erythrocytes in response to a brief period of hypoxia and hypercapnia. Cardiovasc. Res. 1992;26:40–47. doi: 10.1093/cvr/26.1.40. [DOI] [PubMed] [Google Scholar]
- 4.Grierson JP, Meldolesi J. Shear stress-induced [Ca2+]i transients and oscillations in mouse fibroblasts are mediated by endogenously released ATP. J. Biol. Chem. 1995;270:4451–4456. doi: 10.1074/jbc.270.9.4451. [DOI] [PubMed] [Google Scholar]
- 5.Sprague RS, Ellsworth ML, Stephenson AH, Lonigro AJ. ATP: the red blood cell link to NO and local control of the pulmonary circulation. Am. J. Physiol. 1996;271:H2717–H2722. doi: 10.1152/ajpheart.1996.271.6.H2717. [DOI] [PubMed] [Google Scholar]
- 6.Milner P, Kirkpatrick KA, Ralevic V, Toothill V, Pearson J, Burnstock G. Endothelial cells cultured from human umbilical vein release ATP, substance P and acetylcholine in response to increased flow. Proc. Biol. Sci. 1990;241:245–248. doi: 10.1098/rspb.1990.0092. [DOI] [PubMed] [Google Scholar]
- 7.Osipchuk Y, Cahalan M. Cell-to-cell spread of calcium signals mediated by ATP receptors in mast cells. Nature. 1992;359:241–244. doi: 10.1038/359241a0. [DOI] [PubMed] [Google Scholar]
- 8.Smith JP, Haddad EV, Downey JD, Breyer RM, Boutaud O. PGE2 decreases reactivity of human platelets by activating EP2 and EP4. Thromb. Res. 2010;126:e23–e29. doi: 10.1016/j.thromres.2010.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tokunaga A, Tsukimoto M, Harada H, Moriyama Y, Kojima S. Involvement of SLC17A9-dependent vesicular exocytosis in the mechanism of ATP release during T cell activation. J. Biol. Chem. 2010;285:17406–17416. doi: 10.1074/jbc.M110.112417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Abbracchio MP, Burnstock G, Boeynaems JM, Barnard EA, Boyer JL, Kennedy C, Knight GE, Fumagalli M, Gachet C, Jacobson KA, Weisman GA. International Union of Pharmacology LVIII: update on the P2Y G protein-coupled nucleotide receptors: from molecular mechanisms and pathophysiology to therapy. Pharmacol. Rev. 2006;58:281–341. doi: 10.1124/pr.58.3.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Burnstock G, Knight GE. Cellular distribution and functions of P2 receptor subtypes in different systems. Int. Rev. Cytol. 2004;240:31–304. doi: 10.1016/S0074-7696(04)40002-3. [DOI] [PubMed] [Google Scholar]
- 12.Khakh BS, Burnstock G, Kennedy C, King BF, North RA, Seguela P, Voigt M, Humphrey PP. International union of pharmacology. XXIV. Current status of the nomenclature and properties of P2X receptors and their subunits. Pharmacol. Rev. 2001;53:107–118. [PubMed] [Google Scholar]
- 13.Jin J, Daniel JL, Kunapuli SP. Molecular basis for ADP-induced platelet activation. II. The P2Y1 receptor mediates ADP-induced intracellular calcium mobilization and shape change in platelets. J. Biol. Chem. 1998;273:2030–2034. doi: 10.1074/jbc.273.4.2030. [DOI] [PubMed] [Google Scholar]
- 14.Stucky CL, Medler KA, Molliver DC. The P2Y agonist UTP activates cutaneous afferent fibers. Pain. 2004;109:36–44. doi: 10.1016/j.pain.2004.01.007. [DOI] [PubMed] [Google Scholar]
- 15.Yitzhaki S, Shneyvays V, Jacobson KA, Shainberg A. Involvement of uracil nucleotides in protection of cardiomyocytes from hypoxic stress. Biochem. Pharmacol. 2005;69:1215–1223. doi: 10.1016/j.bcp.2005.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bar I, Guns PJ, Metallo J, Cammarata D, Wilkin F, Boeynams JM, Bult H, Robaye B. Knockout mice reveal a role for P2Y6 receptor in macrophages, endothelial cells, and vascular smooth muscle cells. Mol. Pharmacol. 2008;74:777–784. doi: 10.1124/mol.108.046904. [DOI] [PubMed] [Google Scholar]
- 17.Capra V, Ravasi S, Accomazzo MR, Citro S, Grimoldi M, Abbracchio MP, Rovati GE. CysLT1 receptor is a target for extracellular nucleotide-induced heterologous desensitization: a possible feedback mechanism in inflammation. J. Cell Sci. 2005;118:5625–5636. doi: 10.1242/jcs.02668. [DOI] [PubMed] [Google Scholar]
- 18.Chang K, Hanaoka K, Kumada M, Takuwa Y. Molecular cloning and functional analysis of a novel P2 nucleotide receptor. J. Biol. Chem. 1995;270:26152–26158. doi: 10.1074/jbc.270.44.26152. [DOI] [PubMed] [Google Scholar]
- 19.Grbic DM, Degagne E, Langlois C, Dupuis AA, Gendron FP. Intestinal inflammation increases the expression of the P2Y6 receptor on epithelial cells and the release of CXC chemokine ligand 8 by UDP. J. Immunol. 2008;180:2659–2668. doi: 10.4049/jimmunol.180.4.2659. [DOI] [PubMed] [Google Scholar]
- 20.Jiang Y, Borrelli L, Bacskai BJ, Kanaoka Y, Boyce JA. P2Y6 receptors require an intact cysteinyl leukotriene synthetic and signaling system to induce survival and activation of mast cells. J. Immunol. 2009;182:1129–1137. doi: 10.4049/jimmunol.182.2.1129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Marcet B, Horckmans M, Libert F, Hassid S, Boeynaems JM, Communi D. Extracellular nucleotides regulate CCL20 release from human primary airway epithelial cells, monocytes and monocyte-derived dendritic cells. J. Cell Physiol. 2007;211:716–727. doi: 10.1002/jcp.20979. [DOI] [PubMed] [Google Scholar]
- 22.Shin A, Toy T, Rothenfusser S, Robson N, Vorac J, Dauer M, Stuplich M, Endres S, Cebon J, Maraskovsky E, Schnurr M. P2Y receptor signaling regulates phenotype and IFN-alpha secretion of human plasmacytoid dendritic cells. Blood. 2008;111:3062–3069. doi: 10.1182/blood-2007-02-071910. [DOI] [PubMed] [Google Scholar]
- 23.Somers GR, Hammet FM, Trute L, Southey MC, Venter DJ. Expression of the P2Y6 purinergic receptor in human T cells infiltrating inflammatory bowel disease. Lab Invest. 1998;78:1375–1383. [PubMed] [Google Scholar]
- 24.Southey MC, Hammet F, Hutchins AM, Paidhungat M, Somers GR, Venter DJ. Molecular cloning and sequencing of a novel human P2 nucleotide receptor. Biochim. Biophys. Acta. 1996;1309:77–80. doi: 10.1016/s0167-4781(96)00148-0. [DOI] [PubMed] [Google Scholar]
- 25.Tsukimoto M, Tokunaga A, Harada H, Kojima S. Blockade of murine T cell activation by antagonists of P2Y6 and P2×7 receptors. Biochem. Biophys. Res. Commun. 2009;384:512–518. doi: 10.1016/j.bbrc.2009.05.011. [DOI] [PubMed] [Google Scholar]
- 26.Khine AA, Del SL, Vaschetto R, Voglis S, Tullis E, Slutsky AS, Downey GP, Zhang H. Human neutrophil peptides induce interleukin-8 production through the P2Y6 signaling pathway. Blood. 2006;107:2936–2942. doi: 10.1182/blood-2005-06-2314. [DOI] [PubMed] [Google Scholar]
- 27.Warny M, Aboudola S, Robson SC, Sevigny J, Communi D, Soltoff SP, Kelly CP. P2Y(6) nucleotide receptor mediates monocyte interleukin-8 production in response to UDP or lipopolysaccharide. J. Biol. Chem. 2001;276:26051–26056. doi: 10.1074/jbc.M102568200. [DOI] [PubMed] [Google Scholar]
- 28.Riegel AK, Faigle M, Zug S, Rosenberger P, Robaye B, Boeynaems JM, Idzko M, Eltzschig HK. Selective induction of endothelial P2Y6 nucleotide receptor promotes vascular inflammation. Blood. 2011;117:2548–2555. doi: 10.1182/blood-2010-10-313957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Feil S, Valtcheva N, Feil R. Inducible Cre mice. Methods Mol. Biol. 2009;530:343–363. doi: 10.1007/978-1-59745-471-1_18. [DOI] [PubMed] [Google Scholar]
- 30.Lundequist A, Nallamshetty SN, Xing W, Feng C, Laidlaw TM, Uematsu S, Akira S, Boyce JA. Prostaglandin E(2) exerts homeostatic regulation of pulmonary vascular remodeling in allergic airway inflammation. J. Immunol. 2010;184:433–441. doi: 10.4049/jimmunol.0902835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Giannattasio G, Fujioka D, Xing W, Katz HR, Boyce JA, Balestrieri B. Group V secretory phospholipase A2 reveals its role in house dust mite-induced allergic pulmonary inflammation by regulation of dendritic cell function. J. Immunol. 2010;185:4430–4438. doi: 10.4049/jimmunol.1001384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Friend DS, Gurish MF, Austen KF, Hunt J, Stevens RL. Senescent jejunal mast cells and eosinophils in the mouse preferentially translocate to the spleen and draining lymph node, respectively, during the recovery phase of helminth infection. J. Immunol. 2000;165:344–352. doi: 10.4049/jimmunol.165.1.344. [DOI] [PubMed] [Google Scholar]
- 33.Laidlaw TM, Steinke JW, Tinana AM, Feng C, Xing W, Lam BK, Paruchuri S, Boyce JA, Borish L. Characterization of a novel human mast cell line that responds to stem cell factor and expresses functional FcepsilonRI. J. Allergy Clin. Immunol. 2011;127:815–822. doi: 10.1016/j.jaci.2010.12.1101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Vermaelen K, Pauwels R. Accurate and simple discrimination of mouse pulmonary dendritic cell and macrophage populations by flow cytometry: methodology and new insights. Cytometry A. 2004;61:170–177. doi: 10.1002/cyto.a.20064. [DOI] [PubMed] [Google Scholar]
- 35.Triggiani M, Oriente A, Marone G. Differential roles for triglyceride and phospholipid pools of arachidonic acid in human lung macrophages. J. Immunol. 1994;152:1394–1403. [PubMed] [Google Scholar]
- 36.Balestrieri B, Maekawa A, Xing W, Gelb MH, Katz HR, Arm JP. Group V secretory phospholipase A2 modulates phagosome maturation and regulates the innate immune response against Candida albicans. J. Immunol. 2009;182:4891–4898. doi: 10.4049/jimmunol.0803776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rabinowitz SS, Gordon S. Macrosialin, a macrophage-restricted membrane sialoprotein differentially glycosylated in response to inflammatory stimuli. J. Exp. Med. 1991;174:827–836. doi: 10.1084/jem.174.4.827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Barrett NA, Maekawa A, Rahman OM, Austen KF, Kanaoka Y. Dectin-2 recognition of house dust mite triggers cysteinyl leukotriene generation by dendritic cells. J. Immunol. 2009;182:1119–1128. doi: 10.4049/jimmunol.182.2.1119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Barrett NA, Rahman OM, Fernandez JM, Parsons MW, Xing W, Austen KF, Kanaoka Y. Dectin-2 mediates Th2 immunity through the generation of cysteinyl leukotrienes. J. Exp. Med. 2011 doi: 10.1084/jem.20100793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kuperman DA, Huang X, Koth LL, Chang GH, Dolganov GM, Zhu Z, Elias JA, Sheppard D, Erle DJ. Direct effects of interleukin-13 on epithelial cells cause airway hyperreactivity and mucus overproduction in asthma. Nat. Med. 2002;8:885–889. doi: 10.1038/nm734. [DOI] [PubMed] [Google Scholar]
- 41.Wills-Karp M, Luyimbazi J, Xu X, Schofield B, Neben TY, Karp CL, Donaldson DD. Interleukin-13: central mediator of allergic asthma. Science. 1998;282:2258–2261. doi: 10.1126/science.282.5397.2258. [DOI] [PubMed] [Google Scholar]
- 42.Nakanishi A, Morita S, Iwashita H, Sagiya Y, Ashida Y, Shirafuji H, Fujisawa Y, Nishimura O, Fujino M. Role of gob-5 in mucus overproduction and airway hyperresponsiveness in asthma. Proc. Natl. Acad. Sci. U. S. A. 2001;98:5175–5180. doi: 10.1073/pnas.081510898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Foster PS, Hogan SP, Ramsay AJ, Matthaei KI, Young IG. Interleukin 5 deficiency abolishes eosinophilia, airways hyperreactivity, and lung damage in a mouse asthma model. J. Exp. Med. 1996;183:195–201. doi: 10.1084/jem.183.1.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hogan SP, Mould A, Kikutani H, Ramsay AJ, Foster PS. Aeroallergen-induced eosinophilic inflammation, lung damage, and airways hyperreactivity in mice can occur independently of IL-4 and allergen-specific immunoglobulins. J. Clin. Invest. 1997;99:1329–1339. doi: 10.1172/JCI119292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Barrett NA, Austen KF. Innate cells and T helper 2 cell immunity in airway inflammation. Immunity. 2009;31:425–437. doi: 10.1016/j.immuni.2009.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Trompette A, Divanovic S, Visintin A, Blanchard C, Hegde RS, Madan R, Thorne PS, Wills-Karp M, Gioannini TL, Weiss JP, Karp CL. Allergenicity resulting from functional mimicry of a Toll-like receptor complex protein. Nature. 2009;457:585–588. doi: 10.1038/nature07548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hammad H, Chieppa M, Perros F, Willart MA, Germain RN, Lambrecht BN. House dust mite allergen induces asthma via Toll-like receptor 4 triggering of airway structural cells. Nat. Med. 2009;15:410–416. doi: 10.1038/nm.1946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hume DA. Macrophages as APC and the dendritic cell myth. J. Immunol. 2008;181:5829–5835. doi: 10.4049/jimmunol.181.9.5829. [DOI] [PubMed] [Google Scholar]
- 49.Evaldsson C, Ryden I, Uppugunduri S. Anti-inflammatory effects of exogenous uridine in an animal model of lung inflammation. Int. Immunopharmacol. 2007;7:1025–1032. doi: 10.1016/j.intimp.2007.03.008. [DOI] [PubMed] [Google Scholar]
- 50.Muller T, Grimm M, de Vieira RP, Cicko S, Durk T, Sorichter S, Zissel G, Idzko M. Local administration of uridine suppresses the cardinal features of asthmatic airway inflammation. Clin. Exp. Allergy. 2010;40:1552–1560. doi: 10.1111/j.1365-2222.2010.03518.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.